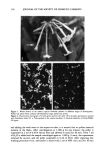
162 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (4) (5) (6) (7) (8) (9) (10) (11) (12) (13) (14) (15) (16) (17) (18) (19) (20) (21) (22) (23) (24) (25) (26) (27) R. M. Cox, Sensitivity of forest plant reproduction to long range transported air pollutants: In vitro sensitivity of pollen to simulated acid rain, New Phytol., 95, 269-276 (1983). R. M. Cox, The sensitivity of pollen from various coniferous and broad-leafed trees to combinations of acidity and trace metals, New Phytol., 109, 193-201 (1988). J. H. B. Wolters and M. J. M. Martens, Effects of air pollutants on pollen, Bot. Rev., 53, 372-414 (1987). Y. Iwanami, Physiological studies of pollen, J. Yokohama Munic. Univ., 116, 1-137 (1959). F. W. Martin, In vitro measurement of pollen tube growth inhibition, Plant Physiol., 49, 924-925 (1972). R. Kappler and U. Kristen, Photometric quantification of in vitro pollen tube growth: A new method suited to determine the cytotoxicity of various environmental substances, Envir. Exp. Bot., 27, 305-309 (1987). R. Kappler and U. Kristen, Photometric quantification of water-insoluble polysaccharides produced by in vitro grown pollen tubes, Envir. Exp. Bot., 28, 33-36 (1988). J. H. Draize, Appraisal of the Saj•ty of Chemicals in Foods, Drugs and Cosmetics--Dermal Toxicity (Association of Food and Drug Officials of the United States, 1959), pp. 46-59. S. D. Gettings, L. C. Dipasquale, D. M. Bagley, M. Chudkowski, J. L. Demetrulias, P. I. Feder, K. L. Hintze, K. D. Marenus, W. J. W. Pape, M. Roddy, R. Schnetzinger, P. M. Silber, J. J. Teal, and S. L. Weise, The CTFA evaluation of alternatives program: An evaluation of in vitro alternatives to the Draize primary eye irritation test (phase I) hydroalcoholic formulations A prelim- inary communication. In Vitro Toxicology, 3, 293-302 (1990). S. D. Gettings, J. J. Teal, D. M. Bagley, J. L. Demetrulias, L. C. Dipasquale, K. L. Hintze, M. G. Rozen, S. L. Weise, M. Chudkowski, K. D. Marenus, W. J. W. Pape, M. Roddy, R. Schnetzinger, P.M. Silber, S. M. Glaza, and P. J. Kurtz, The CTFA evaluation of alternatives program: An evaluation of in vitro alternatives to the Draize primary eye irritation test (phase I) hydroalcoholic formulations (Part 2) Data analysis and biological significance. In Vitro Toxicology, 4, 247-288 (1991). B. Ekwall, and 27 co-authors, Further MEIC evaluation. Acute lethal toxicity in man of reference chemicals Nos. 11-30 predicted by 60 in vitro toxicity assays and by oral LD5o in rodents. Proceedings of lNVITOX Congress, 1992, DeHaan, Belgium. M. Meyberg, R. Kappler, and U. Kristen, "Untersuchungen zur Cytotoxizi6it atmogener Organika des Standortes 'Postturin'," in Das Forschungsprogramm Waldsch•iden am Standort "Postturm," Forstamt Farchau/Ratzeburg, J. Bauch and W. Michaelis, Eds. (GKSS-Forschungszentrum Geesthacht 88/E/55, 1988, pp. 131-137. K. Strube, D. Janke, R. Kappler, and U. Kristen, Toxicity of some herbicides to in vitro growing tobacco pollen tubes (the pollen test), Envir. Exp. Bot., 31, 217-222 (1991). U. Kristen, U. Joos, J. P. van Aken, and R. Kappler, Bestimmung der Zytotoxizi6it von Deter- gentien, Pestiziden und anderen Chemikalien mir dem Pollen-Test (pollen tube growth test), VDI Berichte, 901, 1191-1210 (1991). E. Weber, Grundriss der biologischen $tatistik (Gustav Fischer Verlag, Stuttgart, 1967), pp. 339-342. J. Werner, Medizinische $tatistik (Urban und Schwarzenberg, Munich, Vienna, Baltimore, 1984), pp. 144-146. A. Helenius and K. Simons, Solubilization of membranes by detergents, Biochim. B iophys. Acta, 415, 29-79 (1975). C. Tanford and M. J. Reynolds, Characterization of membrane proteins in detergent solution, Bio- chim. Biophys. Acta, 457, 133-170 (1976). T. K. Surowy and M. R. Sussman, Immunological crossreactivity and inhibitor sensitivities of the plasma membrane H-ATPase from plants and fungi, Biochim. Biophys. Acta, 848, 24-29 (1986). M. I. DeMichelis, C. Olivari, M. C. Pugliarello, and F. Rasicaldogno, Effect ofMg 2+ , Triton X-100 and temperature on basal and FC-stimulated plasma membrane ATPase activity, Plant Sci., 54, 117-124 (1988). K. S. Rao and U. Kristen, The influence of the detergent Triton X-100 on the growth and ultra- structure of tobacco pollen tubes, Can. J. Bot., 68, 1131-1138 (1990). M. K. Kandasamy and U. Kristen, Pentachlorophenol affects mitochondria and induces formation of Golgi apparatus-endoplasmic reticulum hybrids in tobacco pollen tubes, Protoplasma, 141, 112-120 (1987). M. K. Kandasamy and U. Kristen, Influence of triethyl lead on growth and ultrastructure of tobacco pollen tubes, Envir. Exp. Bot., 29, 283-292 (1989). C. S. Weil and R. A. Scala, Study of intra- and interlaboratory variability in the results of rabbit eye and skin irritation test, Toxicol. Appl. Pharmacol., 19, 276-360 (1971).
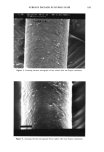
j. Soc. Cosmet. Chem., 44, 163-175 (May/June 1993) A simple and sensitive technique, based on protein loss measurements, to assess surface damage to human hair SUKHVINDER S. SANDHU and CLARENCE R. ROBBINS, Technology Center, Colgate Palmolive Company, Piscataway, NJ 08854-1340. Received October 14, 1992. Synopsis This article describes a simple yet sensitive technique to assess surface damage to hair. It is based on the hypothesis that the damaged hair surface is more susceptible to abrasion/erosion than undamaged hair, and involves shaking hair in water and quantitatively measuring the amount of protein abraded/eroded from the hair using a colorimetric procedure capable of detecting as little as 5 Ixg of protein per mi. With this procedure, we were able to demonstrate significant differences in hair damaged due to bleaching, permanent wave treatments, and Suprox (a diperisophthalic acid based oxidant), and even after extended treatment with different surfactants. Furthermore, hair with exposed cortex was found to be more susceptible to protein loss by surfactants and/or water than hair with intact cuticle. INTRODUCTION Hair fibers generally consist of three distinct morphological components, the outer protective layers known as the cuticle,' the major structural components, or the cortex,' and the porous components, or the medulla. The cuticle plays an important role both as a protective barrier and for many of the cosmetic properties of the hair, whereas the cortex provides mechanical strength to the hair fiber as a whole. It is known that the physi- cochemical properties of hair change as a result of damage to hair (1-3). Damage to hair may be confined to the surface layers (cuticle), or the whole fiber may be involved. However, cuticle layers are generally damaged before any damage can occur to the cortex. Therefore, damage tests that focus more on surface changes have the potential to be more sensitive than those that rely on whole fiber changes. Recently, one of the above authors (Robbins) demonstrated, using Suprox (a diperiso- phthalic acid based oxidant), that extensive damage to several cuticle (surface) layers of hair was readily observed microscopically without any detectable changes in the tensile properties (wet or dry) (4). These findings further support the hypothesis that tensile properties are due primarily to the cortex. Obviously, tensile properties are of little value to study cuticle or surface damage. This report describes a simple and sensitive tech- nique developed to assess surface damage to hair. Some investigators have shown that shaking animal or human hair in the presence of a liquid can be used to separate cuticle 163
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)