I. Soc. Cosmet. Chem., 24, 551-560 (August 19, 1973) Chromatographic Determination of Trace Components in D and C Red No. 37 B. L. KABACOFF, B.S., GEORGE MOHR, and C. M. FAIRCHILD, B.S.* Presented October 9, 1972, Joint Symposium of the Society of Cosmetic Chemists and the Association of O•cial Analytical Chemists, Washington, D.C. Synopsis-Methods of determination of trace components of D AND C RED NO. 37 are presented. PHTHALIC ACID and M-DIETHYLAMINOPHENOL are silylated with N,O-bis(trimethylsilyl) acetamide in the presence of a vast excess of other components of the dye such as Rhodamine B and stearic acid. These other components compete for the trimethylsilyl group. Proper selection of the ratio of the silylating agent to solvent and catalyst results in a linear response upon subsequent GAS-LIQUID CHROMATOGRA- PHY. o-(2-Hydroxy-4-diethylaminobenzoyl) benzoic acid is readily separated from the other constituents of the dye by THIN-LAYER CHROMATOGRAPHY. The band con- taining this material is eluted from the tic plate and read, in a spectrophotometer. This substance may also be separated from the other components by SILYLATION and high- temperature glc. Triethylrhodamine may also be assayed by tic. In the solid skate it is apparently unstable. INTRODUCTION The object of the study reported here was the development of simple rapid methods for the determination of phthalic acid, N, N-diethyl-m-aminophenol, o-(2-hydroxy-4-diethylaminobenzoyl) benzoic acid (HDBA), and triethyl- rhodamine in D and C Red No. 37 (Rhodamine B stearate). Phthalic acid has previously been assayed by extraction by immiscible solvent, followed by spectrophotometric reading in D and C Red No. 19 (Rhodamine B HC1) and other dyes (1, 2). Diethylaminophenol has been assayed in both D and C Red Nos. 19 and 37 by steam distillation followed by spectrophotometric reading (3). In the method described below, both substances are determined simul- taneously by gas-liquid chromatography. *Revlon Research Center, Inc. 945 Zerega Ave., Bronx, N.Y. 10473. 551
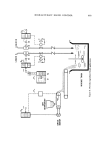
552 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS HDBA in D and C Red No. 19 has been assayed by column partition chromatography followed by spectrophotometric monitoring of the eluates (4). The method describcd below utilizes thin-layer chromatography to sepa- rate this compound from the other components of the dye. Triethylrhodamine in D and C Red No. 19 has been isolated from the other components of the dye by the same partition chromatographic technique (4). This paper does not report quantitative assay of triethylrhodamine. The authors have separated triethylrhodamine by thin-layer chromatography but have found that the compound is quite unstable in the solid state. It was necessary, therefore, to resort to silica gel column chromatography to determinc the purity of the triethylrhodamine standard. EXPERIMENTAL Apparatus and Reagents A gas-liquid chromatograph* was used with a flame ionization detector. The glc column used for the determination of phthalic acid and m-diethyla- minophenol was a 10-ft x 0.25-in. coppcr column contain ,ng 5% OV 11t on Chromosorb WAW 60/80 mesh.* Septumsõ (0.5 in. in diameter) were used to cap the silylation vials with the Teflon coating facing the vial contents. They are also used in the injection ports to elimlnate spurious peaks at high temperatures. Silylation vials were made by drilling a 1/4-in. hole in the cap of a 1/s-.oz. flint glass bottle. I I The septum described above is placed in the cap. Any similar container of comparable size whose cap will hold the septum snugly can be used. For thin-layer chromatography, the tlc platesô were 20 x 20 cm for HDBA assays and silica gel G (F254)** for the triethylrhodamine assay. The tlc migrating tank can be any one suitable for use with 20 x 20 cm plates. A tic spot collector (No. 0410 130-8)** and 25-ml standard taper (14/20) flasks were used. The sample applicator*** was a threaded plunger syringe. For filtered ultraviolet light, any uv light emitting at 254 and 365 nm, such as UVSL-55 Mineralight, # can be used. The silylating mixture for the assay of phthalic and diethylaminophenol was prepared by mixing equal volumes of N,O-bis (trimethylsilyl) acetamide and * Model 5750, Hewlett Packard, Palo Alto, Calif. 94304. ? Supelco, Bellefonte, Pa. 16823. * Johns Manville, Celite Div., New York, N.Y. 10016. õ No. F-138, Canton Bio-Medical Products, Swarthmore, Pa. 19081. [[ Cat. No. A 6710, J. Rabinowitz & Sons, Brooklyn, N.Y. •T Quantagram LQIF, Quanttun Industries, Fairfield, N.J. 07006. ** Brinkmann Instruments, Inc., Westbury, N.Y. 11590. *** Model 87001, Hamilton Co., Whittier, Calif. 90608. •p Ultraviolet Products, Inc., San Gabriel, Calif. 91778.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)