PROPERTIES AND STRUCTURE OF SILICAS 499 TYPE SILICAS I Synthetic Silicas I I Vapor Liquid Process Process 1 Silica Gel Precipitated Silica , I Figure 2. Type silicas Xerogel B. TYPES OF SILICAS A system for classifying the various types of synthetic silicas is found in Figure 2. It is shown in Figure 2 that silica gels are of two types, xerogels and aerogels. During the manufacture of silica gels first a hydrosol, then a hydrogel is formed. When a hydrogel is washed and then dried from an organic medium without the shrinkage of structure (4), it is called an aerogel. Otherwise, it is called a xerogel. Development of new, controlled-structure silicas has created the need for further classifying the precipitated silicas into five classes (see Figure 2): VHS--Very High Structure, HS--High Structure, MS--Medium Structure, LS--Low Structure and VLS--Very Low Structure. The end-use properties of a precipitated silica are related to its structure, its particle size and its surface silanol group density. The definitions of the VHS to VLS are dis- cussed later in the text under Section III-B. C. COMPARATIVE PROPERTIES OF SYNTHETIC SILICAS The properties of the fumed silicas (prepared via the vapor process) have been studied by many workers (5-7) and their properties have been found to be similar in some respects to the silicas prepared by the liquid phase or wet process. This is understand- able in that the properties of the fine-particle synthetic silicas, regardless of their method of preparation, must be related to the following key parameters: BET surface area, particle size and silanol group density.
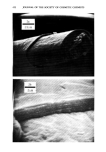
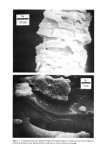
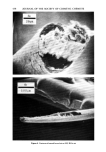
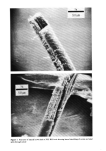
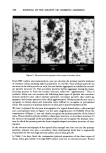
500 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS •ILICA P•ECIPITATE•D SILICA Figure 3. Electron photomicrographs of three types of synthetic silicas From BET surface area measurement, one can calculate the primary particle diameter of synthetic silicas assuming spherical particles. In real life, these primary particles do not exist in the silica powder as such, but are further aggregated to exhibit the second- ary particle structure (3). The secondary particles further aggregate during the manu- facturing process to form the tertiary structure called the "agglomerates." Thus in synthetic silicas one can envision the following three types of particle size structure: primary particles (also called ultimate particles), secondary particles (also called ag- gregates) and tertiary particles (also called agglomerates). Primary particles are easy to recognize in fumed silicas and somewhat more difficult to recognize in precipitated silicas. The existence of primary particles in silica gels is purely hypothetical (8). We have compared the electron micrographs of a typical fumed silica, a silica aerogel and a precipitated silica in Figure 3 at the same magnification of 69,800X. From the examination of Figure 3, one can clearly recognize the primary particles of the fumed silica. These primary particles exhibit a chain-type structure or secondary structure. In the electron micrograph of the precipitated silica one can recognize the primary struc- ture in which the primary particles form irregular-shaped secondary structure of vary- ing sizes. In the electron micrograph of the typical silica aerogel one does not see the primary particles instead, one sees a secondary, three dimensional mesh that is supposedly responsible for the very high internal surface area in silica gels (8). In Table I we have listed the comparative physical properties of the three types of silicas. Silica gels exhibit very high BET surface area due to internal porosity. Fumed
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)