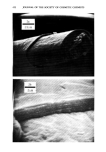
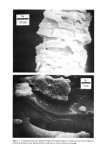
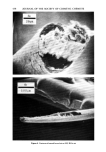
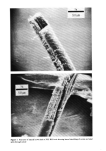
PROPERTIES AND STRUCTURE OF SILICAS 519 Table VIII Cosmetic Application of Silicas PROPERTY DRYING-UP LIQUIDS ANTI-CAKING CONDITIONING AGENT SUSPENSION THICKENING TABLETTING AID ABRASIVES AND POLISHING AGENT ADSORBENT VISCOSITY CONTROL GLOSS REDUCTION APPLICATION FOODS, COSMETICS, PHARMACEUTICALS FOODS, COSMETICS, HOUSEHOLD PRODUCTS PERSONAL CARE, SPECIALTY HAIR CARE, DEODORANTS, COSMETICS, LIQUIDS TOOTHPASTE, LIQUIDS, SPECIALTY PHARMACEUTICALS, COSMETICS TOOTHPASTE POLYOLS NAIL POLISHES, COSMETICS SPECIALTY SILICA STRUCTURE VHS, HS VHS, HS VHS VHS VHS, MS VHS, MS LS, VLS LS, VLS VHS VHS, MS B. ANTI-CAKING Many cosmetic, food and pharmaceutical preparations tend to cake up in the container or the box in which they are packaged. Due to the fine particle size and very high absorption capacity of VHS or HS silica, these silicas are widely used to make cosmetic and specialty formulations free-flowing. C. CONDITIONING Some cosmetic and specialty powder formulations are difficult to flow due to moisture pick-up by one or several ingredients of the formulations. The flow properties and angle of repose of these powder formulations can be improved in such situations by the addition of 1 to 2% by weight of synthetic silica. D. SUSPENSION AID In many cosmetic formulations, such as in aerosol and antiperspirant formulations, a very small amount of silica is needed to provide suspension characteristics to the active ingredients of the formulation. A VHS or HS silica of high absorption capacity and sur- face area is used in this type of application. E. THICKENING AGENT The addition of 6 to 16% VHS or HS silicas to dentifrice formulations, aqueous or oil systems, would convert them to gels or pastes. The superior thickening efficiency of a precipitated silica depends upon its structure, particle size, surface area and nature of additives. r. TABLETTING AID Precipitated silicas serve as binders, eliminate sticking and act as tabletting aids in phar- maceutical and specialty applications.
520 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS G. ABRASIVE AND POLISHING AGENT Due to mild abrasivity and controlled structure, precipitated silicas are used as mild abrasives and polishing agents in toothpaste formulations. LS and VLS silicas are ideally used because of their low humectant demand and acceptable compatibility features with other toothpaste formula ingredients. H. ADSORBENT Controlled-structure precipitated silicas are used to purify polyols and to remove traces of impurities from many liquids and effluents. The choice of proper silica would depend upon the nature and concentration of the impurity present in the liquid. I. VISCOSITY CONTROL A relatively small amount of VHS silicas increases the viscosity of creams, lotions, oint- ments, ache preparations and other specialty products enabling the cosmetic chemist to control the viscosity characteristics of his finished product. J. GLOSS REDUCTION Synthetic silicas are widely used as de-lustering agents to reduce the gloss of varnishes, lacquers and other surface coatings. VII. CONCLUSIONS We have presented here the structure, chemistry and preparation of controlled-struc- ture precipitated silicas. We have correlated the properties and the end-use applica- tions of silicas to their structure. The variety of cosmetic applications in which synthetic silicas are currently being used is already phenomenal. Further insight into surface chemistry and silica structure is needed to further explore the future cosmetic properties of synthetic silicas. REFERENCES (8) (9) (10) (1) CTFA Cosmetic Ingredient Dictionary, Bulletin No. 3 (March 1974), published by the Cosmetic, Toiletry and Fragrance Association, Washington, D.C. (2) E. Wagner and H. Brunner, Angew. Chem., 72,744 (1960). (3) R. Bode, H. Ferch and H. Fratzscher, Kaut. Gummi Kunstst., 20, 578 (1967). (4) R. K. Iler, "The Colloid Chemistry of Silica and Silicates," The Cornell University Press, Ithaca, New York, 1955, Chapter VI. (5) A. V. Kiselev, "The Structure and Properties of Porous Materials," Butterworths Scientific Publica- tions, London, England, 1958, p 158. (6) J. A. Hockey and B. A. Pethica, Surface hydration of silicas, Trans. Faraday Soc., 57, 2247 (1961). (7) J. A. Hockey, "The Nature of the Silica Surface," Ph.D. Thesis, Manchester University, Manchester, England, 1960. F. Kindervater, Concerning the structure of silica fillers, Kaut. Gummi Kunstst., 26, 7 (1973). J. A. Hockey, The surface properties of the silica powder, Chem. Ind. (London), January, 57 (1965). M. L. Hair, "Infrared Spectroscopy in Surface Chemistry," Marcel Dekker, Inc., New York, New York, 1967, Chapter 4.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)