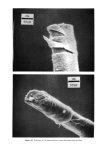
PROPERTIES AND STRUCTURE OF SILICAS 515 50,000 10,000 1,000 EFFECT OF SILICA CONCENTRATION ON VISCOSITY OF MINERAL OIL Sl LICA A = VHS SILICA B = HS " C = MS " D = MS " E = LS " F = VLS " AA = SAMPLE A CALClNATED @ (600øC) iAA ,D I I I I I 1.0 2.0 4.0 6.0 8.0 10 CONC. OF SILICA (%) Figure 18. Effect of silica concentration on viscosity of mineral oil From Figure 18, it is clear that only 3 to 6% of VHS silica (Sample A) is extremely ef- fective in increasing the viscosity of mineral oil. At the same level of concentration (3 to 6%) the viscosity increase produced by the VHS silica (Sample A) is much lower in the glycerine medium. This result is explained by the fact that the precipitated silicas produce a much higher viscosity increase when added to the nonhydrogen-bonding liquid system (mineral oil). In the nonhydrogen-bonding systems, the hydroxyl groups of the precipitated silicas have a greater tendency .to hydrogen bond with each other because there is no com- petition for hydrogen bonding in the liquid medium. This situation is reversed when the silica is added to a hydrogen-bonding liquid sLystem such as glycerine. The hy- drogen-bonding liquid system competes for the silanol group of silicas thereby reduc- ing the ability of the silica-silanol groups to hydrogen bond with each other. This , explains the lower viscosity increase observed when silicas are •ad.ded ,to the hydrogen- bonding liquid system. The viscosity of a liquid system xvould increase when the silica structure •level increases, the particle size decreases, the BET surface area increases and the silica concentration or use level increases. It appears that the sitanol group density of silicas also influences the viscosity of liquid systems. To prove this concept, a VHS silica, Sample A, was calcined for 2 hr at 600øC to partially remove the silanol groups. This calcined silica material, AA, was added to mineral oil t,o study the viscosity effects. From Figure 18, it is clear that ,rhe viscosity increase contributed by •Sample AA (calcined VHS silica) is much lower than the •vis-
516 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 5000[ 2000 EFFECT OF SILICA CONCENTRATION ON VISCOSITY OF GLYCERINE A: VHS SILICA B: HS " C: MS " F: VLS " AA: SAMPLE A CALCI NED @ (600øC) A ,AA 1000 9OO 8OO 7OO 6OO 2.0 4.0 6.0 8.0 CONC. OF SILICA {%) Figure 19. Effect of silica concentration on viscosity ofglycerine I 10.0 cosity increase obtained with the regular Sample A. This substantiates the fact that silica thickener efficiency is reduced when silanol groups are partially removed. Additives have an effect on the viscosity behavior of silicas in various systems. In general, addition of 1 to 2% by weight (based on total weight of system) of a hydrogen- bonding additive (ethylene glycol, amines, etc.) to precipitated silica in a nonhydrogen- bonding or slightly polar system will increase viscosity because addition of the additive will increase the probability of silanol--silanol group interactions by acting as bridging groups. Large addition of additives (5 to 10%) of hydrogen-bonding additive would tend to decrease the viscosity of nonhydrogen-bonding, liquid-silica system. It has been observed that the most favorable pH range for thickening liquid systems capable of hydrogen bonding is between 4 to 8 the maximum viscosity is obtained with silica close to pH 7. In summarizing, for cosmetic applications one needs to choose a very high structure silica with the highest BET surface area and the finest particle size to obtain the maximum viscosity increase in a given system. B. DENTIFRICE SILICA ABRASIVES VERSUS RDA Dentifrice compositions contain an abrasive, liquid humectant, thickener, sudsing agent, flavor and minor amounts of other optional ingredients. Extensive studies have
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)