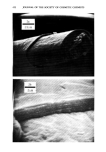
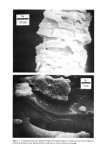
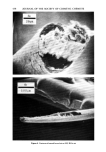
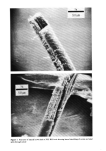
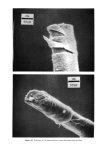
PROPERTIES AND STRUCTURE OF SILICAS 505 VH• •E½IPITATED SILICA •,•OX Figure 7. Electron photomicrographs of VHS and HS silicas I•A•OX Figure 8. Electron photomicrographs of MS and LS silicas
506 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table III Particle Size of Precipitated Silicas PARTICLE DIAMETERS, nm MEAN MINI- MAXI. STRUCTURE EMSA m:/g PARTICLE DIAMETER MUM MUM VHS 250 12 5 20 HS 175 17 5 30 MS 145 20 5 25 LS 60 51 5 300 IV. RESULTS AND DISCUSSIONS A. METHOD OF EVALUATION AND PROPERTIES OF PRECIPITATED SILICAS Six controlled-structure silicas (structure level from VHS to VLS) were prepared similar to the method illustrated in Figure 6. All the precipitated samples were washed, dried and air micronized to produce micron-size silicas. These six samples were designated as A, B, C, D, E and F. The finished samples were characterized by the various physical properties, such as oil absorption (OA), glycerine absorption (GA), mineral oil absorption (MA), sorbitol absorption (SA), BET surface area (BET), average particle size (APS), porosity by mercury intrusion (HGI), glycerine demand index (GDI), mineral oil demand index (MDI), sorbitol demand index (SDI), % wet cake moisture (WCM) and structure index (SI). The data are summarized in Tables IV and V. The oil absorption of silicas was determined by the ASTM rub-out method (16). This test is based on the principle of mixing linseed oil with a silica by rubbing with a spatula on a smooth surface until a stiff putty-like paste is formed. By measuring the quantity of oil required to produce a very stiff, putty-like paste, which will not break or separate when it is cut with the spatula, one can measure the oil absorption of silica--a value which represents the volume of oil required per unit weight of silica to saturate the silica sorptive capacity. The oil absorption value is calculated by the following eq 3. ml oil absorption x 100 Oil Absorption = (3) weight of silica sample, gram = ml oil/100 g silica The ASTM method was modified in that the linseed oil was replaced by glycerine, mineral oil (saybolt viscosity 340 to 350) or sorbitol (70% solution) when running the respective humectant and mineral oil absorption values. Surface area was determined by the nitrogen adsorption method (17) of Brunauer, Em- mett and Teller (BET) while the average particle size (secondary particle size) was de- termined by the Coulter Counter method (18) using Model TAIl. The void volume of silicas was determined by the Aminco-Winslow Porosimeter (19). This instrument is a completely hydraulic machine used to measure the void structure of various materials. The mercury is forced into the voids as a function of pressure and the volume of mercury intruded per gram of sample is calculated at each pressure set- ting. Increments in volume (cc/g) at each pressure setting are plotted against the void size corresponding to the pressure setting increments. We have listed in Table IV the total intruded volume of mercury in cc Hg/g of silica.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)