j. Soc. Cosmet. Chem., 33, 169-178 (July 1982) The mechanical spectra of skin, II. The thermal dependence of the low-strain viscoelastic properties RUSSELL O. POTTS and MIKLOS M. BREUER, Gillette Research Institute, 1413 Research Boulevard, Rockville, MD 20850. Received October 5, 1981. Synopsis We have investigated the low-strain viscoelastic properties of excised hamster skin in order to evaluate the underlying relaxation mechanisms. The stress relaxation in the physiologically important low-strain region was measured and the data transformed into a viscoelastic spectrum. Each spectrum represents H(r), the relaxation modulus as a function of r, the relaxation time constant. Both quantities are characteristic of relaxation mechanisms involved in the viscoelastic response. Relaxation spectra of excised hamster skin at various relative humidities and temperatures show, under all conditions tested, spectra consisting of three peaks (A, B and C) with time constants at 21øC and 95% r.h. near 2, 40 and 300 sec, respectively. For all three peaks r values show only temperature dependence, while the H(r) values vary only with relative humidity. The temperature dependence of r yields activation enthalpies (AH*) for the three peaks. The values of AH* for peaks B and C are in the range of 7-10 Kcal/mole, while peak A has a AH* value near zero within experimental error. On the basis of these results, the values of r and the variation of H(r) with relative humidity, we propose a molecular model for the mechanisms responsible for the mechanical relaxation. The model suggests that the stretching of elastin and subsequent shearing of the mucopolysaccharide-water gel are responsible for the low-strain viscoelastic response. INTRODUCTION Mammalian derreal tissue is highly complex in its function, composition and structure. It serves as the first line of defense against water loss and external shock, both biological and physical. In this last regard the skin is truly remarkable, repeatedly withstanding stresses of hundreds of pounds per square inch and yet remaining soft and pliant. Skin is a layered composite tissue primarily composed of fibrous proteins and gel-like mucopolysaccharides (1). The outermost layer of the epidermis, the stratum comeurn, comprises a relatively small portion of the intact skin and is the ultimate result of the differentiation and outward migration of underlying cells. It is primarily composed of the protein keratin which can exist in highly structured forms stabilized by hydrogen bonding (2). Beneath the epidermis lies the main body of the skin, the derreal tissue. The primary 169
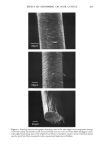
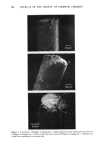
170 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS proteins of this layer are collagen and elastin accounting for approximately 70% and 4% of the dry weight, respectively. Collagen fibers run throughout the skin and are made up of smaller tropocollagen molecules which form hydrogen bonded helices (3). In marked contrast, elastin contains many amorphous regions. About 90% of the amino acids have nonpolar side chains which stabilize the molecule by hydrophobic interactions (4). Furthermore, individual elastin fibers are covalently cross-linked forming a three dimensional network. The dermal protein fibers are embedded in a gel matrix composed of mucopolysac- charides and water, often called the ground substance. The mucopolysaccharides are high molecular weight chains of carbohydrates with covalently attached protein side chains. These macromolecules carry a large negative charge at neutral pH. Due to electrostatic repulsions along the polymer backbone, these molecules probably exist in an extended, stiff conformation under physiological condition (5,6). The elastic (time-independent) properties of skin have been the subject of many investigations (see reference 1 for a review). The results of these experiments indicate that at low strains (20%), the stress varies linearly with strain giving an elastic modulus similar in value to that obtained for pure elastin (approximately 106 dyne/cm2). At high strains, linear behavior is again observed, however, with an elastic modulus several orders of magnitude larger and very similar in magnitude to that obtained for pure collagen. Thus, elastin appears responsible for elastic behavior of skin at low strains while collagen gives skin high tensile strength at large extensions (1). Dermal protein fibers are easily visible by light microscopy. Upon microscopic inspection the fibers exhibit a regular, wavy pattern. The results of microscopy and x-ray diffraction studies suggest that collagen fibers are folded periodically producing the wavy pattern. Increasing strain serves to align the collagen and hence, the waviness vanishes (7). Several authors have suggested similar structural models for skin consistent with the observed properties (7,8). This model suggests that collagen and elastin are cross-linked at regular intervals. Tension exerted by elastin causes the collagen fibers to fold in a repeated manner. Low levels of stretching extends the elastin and aligns the collagen, while at high strain levels both the collagen and elastin molecules become extended. The time-dependent, viscoelastic properties of skin have not been extensively investi- gated. Early indications of the viscoelastic nature of skin were obtained from the results of in vitro stress relaxation experiments of Kenedi et al. (9) showing relaxation time constants over the range of 0.29 to 1700 sec. Lanir and Fung (10) used stress relaxation and hysteresis techniques to investigate directional differences in time-dependent properties of rabbit skin at large strain. Their results showed that the viscoelastic properties varied markedly along orthogonal directions of stretching. Since collagen fibers exhibit preferential alignment, the authors suggested that the large-strain, time-dependent properties were associated with the stretching of collagen fibers. More recently Finlay (11) investigated the torsional properties of skin at low strain, by measuring the phase lag between the periodic displacement and resultant torque over a range of frequencies. His results showed that, over the range of 0.04 to 1 Hz, the phase angle was independent of frequency, a viscoelastic behavior similar to that of mucøpølysaccharide-water gels. Thus, Finlay concluded that the ground substance matrix was responsible for the viscoelastic properties of skin.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)