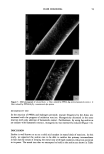
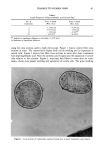
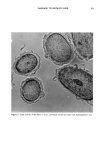
j. Soc. Cosmet. Chem., 42, 1-17 (January/February 1991) Substantivity of dyes and surfactants containing isothiuroniurn groups to hair THOMAS M. DEPPERT, BRYAN P. MURPHY, and JANUSZ Z. JACHOWICZ, Clairol Research Labs, 2 Blachley Road, Stamford, CT 06922. Received August 8, 1990. Presented at the 16th IFSCC Congress, New York, October 1990. Synopsis Reports in the patent and scientific literature have shown that the isothiuronium group is useful for the fixation of dyes to textiles. This technology has seen minimal exploitation in the personal care industry, although it might be viewed as useful in making cosmetic treatments (e.g., dyes and conditioners) more durable. We have tested this hypothesis by preparing a number of dyes and surfactants containing the isothiuronium moiety, and assessing their efficacy in hair treatments under acidic, basic, oxidative, and reductive conditions. The evaluations of dye and surfactant substantivity were performed by the measurements of Hunter tristim- ulus and work of combing values, respectively, before and after shampooing. The adsorption kinetics of model isothiuronium surfactants and the cross sections of hair treated with isothiuronium dyes were also evaluated. In the case of treatments with surfactants, the most pronounced effects were obtained for the diisothiuronium derivative when 1) it was exposed to cysteine residues in reduced hair, followed by oxidation, and 2) the partially hydrolyzed surfactant (i.e., containing free thioIs) was reacted with un- treated hair. The most substantive dyeouts were observed for treatments performed under similar condi- tions. These results suggest that there may be a chemical reaction of the isothiuronium compounds with keratin, although the contribution of electrostatic and Van der Waals interactions cannot be excluded. INTRODUCTION The use of reactive compounds for chemical modification of keratin fibers has been explored in a number of papers. S-alkylation, following fiber reduction, with alkylha- lides or alkyldihalides (1) and N-alkylmaleimides (2), takes advantage of the presence of reactive thiol groups in the fiber structure. These treatments increase the wet mechan- ical properties of the reduced keratin fibers. Deem et al. (3) reported that the reaction of amino, hydroxyl, and thiol groups of hair keratin with phenyl isocyanate, ninhydrin, and formaldehyde improves the fiber wet strength. Patent literature describes the ap- plication of 3-mercapto-2-hydroxypropyl trialkyl ammonium salts (4) as reducing agents for hair, and hexamethyldisilazane (5) for stabilization of alkali-damaged hair (by silylating hydroxyl and amino groups). Another route for keratin modification involves the treatment of weakened hair with reactive silanes such as alkyltrialkoxysilanes in the
2 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS presence of water and alcohol (6). These treatments are claimed to give the hair good settability and curl retention. The improvement of set stability of wool fabrics was also achieved by reacting the wool with Bunte salt containing polymers (7). The area of reactive dyes for wool has been explored for over 20 years. The dyes with reactive moieties assure a high substantivity to wool protein, and consequently wash- fastness of the dyeouts. Currently, reactive dyestuffs containing ot-bromoacrylamido, 2,4-difiuoro-5-chloropyrimidyl, and [•-sulphato- or N-methyltaurine-ethylsulfone groups are commercially available (8). These compounds are, however, too reactive and probably not safe enough to be considered for use in consumer products such as hair dyeing compositions. In 1975 Guise and Stapleton showed that the isothiuronium group could be useful for the fixation of dyes to wool textiles (9, 10). The process involves the absorption of water-soluble dye into the fiber at neutral pH, high pH hydrolysis of the isothiuronium to the mercapto group, followed by the thiol-disulfide exchange reaction with the fiber protein, or oxidation with the formation of water-insoluble, disulfide-linked dimeric dye molecules or a keratin-dye product. A similar mechanism of binding of thiol or disulfide-containing dyes to wool was reported by Asquith et al. (11,12). A review of the subsequent patent literature revealed the use of isothiuronium compounds as reac- tive dyestuffs for cellulose and wool (13). This technology has not been extensively used in the personal care industry (14), although it can be potentially useful in making cosmetic treatments such as dyes and conditioners more durable. The purpose of this investigation was to ascertain the effect of incoporation of the isothiuronium moiety into cationic surfactants and dyestuffs on their durability (measured by resistance to shampooing) as dyeing and conditioning agents. EXPERIMENTAL MATERIALS Samples of hair. The hair used in this work was purchased from DeMeo Brothers, New York. The hair used in the adsorption and combing experiments was commercially blended virgin brown hair. It was washed with 5% Triton X-100 (Rohm and Haas) and thoroughly rinsed under running deionized water. Hair samples were then soaked in three 500-ml water baths for 15 min each to insure removal of residual surfactant. Hair ø C swatches were dried in vacuo at 37 , and chopped into 2-4-mm-length pieces for use in adsorption experiments. The hair used in dye evaluation experiments was commercially blended gray or pied- mont hair. The blended gray hair was characterized by Hunter tristimulus values of L = 30.36, a = -0.29, andb = 5.47. Dyes (Scheme I). HC Yellow 2 (2 2-(2-nitroanilino)ethanol). Ten grams (70.9 mmol) of 2-nitrofluorobenzene [1] was stirred in 50 g of 2-aminoethanol at 80øC under argon until the starting material was completely reacted. The reaction mixture was poured onto ice, filtered, and recrystallized from 2-propanol to give 10.6 g (82%) of 2. 1-Mesyloxy-2-(nitroanilino)ethane [3]. Compound 2 (10 g 54.9 mmol) was dissolved in 150 ml of anhydrous THF and 5.6 g of dry triethylamine. Methanesulfonyl chloride (6.3 g 55.5 mmol) was added dropwise, while cooling the reaction vessel in an icebath.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)