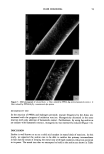
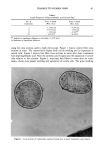
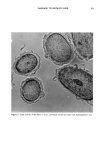
32 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Figure 14a. SI Figure 14b. Figure 14. Scanning electron micrographs of film formed by freeze-drying a 1% Polyquaternium-10 (JR30M), 0.1% SDS composition.
POLYMER/SURFACTANT GELLING STRUCTURES 33 c) Adding a polycation with little tendency to gel with the anionic surfactant but which will compete for the latter. We found that addition of a typical vinyl-based polycation, viz., Polyquaternium-7, led to pronounced thinning of gels, as did the addition of the lowest molecular weight grade of Polyquaternium-10 (JR125). In both cases, how- ever, considerable excess of added polycation was required. FORMULATION IMPLICATIONS In practice, the gel would usually incorporate other ingredients. If they are soluble (and compatible), formulating them into gel compositions of the type described here should present little difficulty. If, on the other hand, the ingredient is insoluble, while the high consistency of the gels would provide stability to settling (or levitation), this same factor would render physical incorporation of the ingredient difficult. For this reason it should be incorporated into one or other of the "master" solutions (polymer or surfac- rant) prior to blending to fabricate the gel. We tested this principle by investigating finely divided alumina and titanium dioxide (representative of positively charged and negatively charged solids, respectively) and a stabilized oil emulsion as model ingre- dients. The latter were dispersed by stirring into either the polymer phase or the surfac- rant phase, followed by carefully blending these two phases--all at room temperature. In all cases, although there were some differences in the appearance of the gel suspen- sions, both procedures led to the formation of stable systems. CONCLUSIONS 1. Strong, clear aqueous gels can be formed with mixtures of high-molecular-weight grade Polyquaternium-10 (JR30M) and a variety of anionic surfactants in the pre- precipitation range. 2. The gels can be made at quite low concentrations of polymer ("• 1%) in mixture with the surfactant at about one tenth of this concentration. 3. Characterization, involving oscillatory measurements, yielded values of the various rheological parameters, of which the storage or elastic modulus (G') is the most useful measure of gelling. 4. In a homologous series of cationic cellulosic polymers, blended with SDS at the optimal gelling ratio, G' increased (a) with polymer molecular weight and (b) with the degree of cationic substitution. 5. In mixtures with the highest molecular weight polymer, gelling efficiency followed the order SDS ES-3 • ES-2 DDBS • AOT. 6. The gels appear to offer interesting formulation opportunities. ACKNOWLEDGMENTS The assistance of K. B. Reuter and J. Petruzzello in obtaining the electronmicrographs is gratefully recognized. REFERENCES (1) E. D. Goddard, Polymer-surfactant interaction. Part I. Uncharged water soluble polymers and charged surfactants. Part II. Polymer and surfactant of opposite charge, Colloids Surfaces, 19, 255-300 and 301-329 (1986).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)