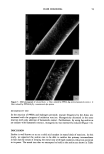
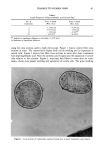
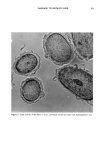
DYES AND SURFACTANTS FOR HAIR 17 (9) (10) (11) (12) (13) (14) (15) (16) (17) (18) (19) (20) (21) Conference on the Chemistry and Application of Reactive Dyes, University of Leeds, September 18-19, 1989. G. B. Guise and I. W. Stapleton, New approaches to washfast dyes for wool. I. Isothiuronium salts as protected dye reactive groups. J.S.D.C., 91, 223 (1975). G. B. Guise and I. W. Stapleton, New approaches to washfast dyes for wool. II. Synthesis and dyeing properties of model isothiuronium dyes, J.S.D.C., 91, 259 (1975). R. S. Asquith and A. K. Puri, Disulphide exchange as a method of coloration of animal fibers, J.S.D.C., 87, 116 (1971). R. S. Asquith, P. Carthew, and T. T. Francis, The reactions of disulphide dyes for the covalent coloration of keratin, J.S.D.C., 89, 168 (1969). US 3,272,794, assigned to American Aniline Products Inc. US 3,803,323, assigned to l'Oreal. R. L. Shriner, R. C. Fuson, D. Y. Curtin, and T. C. Morrill, The Systematic Identification of Organic Compounds, 6th ed. (John Wiley & Sons, New York, 1980), p. 332. J. March, Advanced Organic Chemistry, 3rd ed. (John Wiley & Sons, New York, 1985), p. 220. M. L. Garcia and J. Diaz, Combability measurements on human hair, J. Soc. Cosmet. Chem., 27, 379 (1976). J. A. Swift and A. C. Brown, The critical determination of fine changes in the surface architecture of human hair due to cosmetic treatment, J. Soc. Cosmet. Chem., 23, 695 (1972). S. K. Han, Y. K. Kamath, and H.-D. Weigman, Diffusion of semipermanent dyestuffs in human hair, J. Soc. Cosmet. Chem., 36, 1 (1985). P. Finkelstein and K. Laden, The mechanism of conditioning of hair with alkyl quaternary am- monium compounds, Appl. Polym. Symp., 18, 673 (1971). C. Robbins, C. Reich, and J. Clarke, Dyestaining and the removal of cationics from keratin: The structure and influence of the washing anion, J. Soc. Cosmet. Chem., 40, 205 (1989).
j. Soc. Cosmet. Chem., 42, 19-34 (January/February 1991) Novel gelling structures based on polymer/surfactant systems E. D. GODDARD, P. S. LEUNG, and K. P. A. PADMANABHAN, Union Carbide Chemical and Plastics Company, Inc., Specialty Chemicals Division, Tarrytown, NY 10591. Received A, ugust 15, 1990. Synopsis Our objective was to seek and characterize new gelling compositions based on suitably chosen polymer/sur- factant pairs. In earlier work we reported unusual increases in viscosity when an anionic surfactant, such as SDS, was added to a cationic cellulosic polymer (Polyquaternium-10, medium MW). We have now found even greater increases in apparent viscosity and, indeed, formation of gels of good strength and clarity when a high-molecular-weight version of PQ-10 is used in combination with SDS. Gels are viscoelastic materials, i.e., possess both liquid and solid characteristics and are most appropriately characterized by oscillatory rheology measurements that yield such parameters as storage modulus (G'), loss modulus (G"), phase angle (8), and so on. Measurements, with data analysis, were carried out using the Bohlin VOR Rheometer. The results are interpreted in terms of the formation of "super" macromolecules through association of the alkyl groups of surfactant molecules bound to the ionic sites of different poly- meric molecules by strong electrostatic forces. It is shown that strong gels can be formed at quite low levels of polymer (--1%) and that there is an optimal level of SDS and other anionic surfactants to achieve maximum gel strength. Adjustment of the gel's properties, including liquefaction, can be achieved by suitable adjustment of the surfactant/polymer ratio or the addition of selected cationic materials. Preliminary information relevant to formulation is provided. INTRODUCTION In a series of papers we have drawn attention to the association tendency of water-sol- uble polymers and surfactants in aqueous solution. The various factors involved in the association reactions, and their analysis, have been the subject of recent reviews [see, for example, references (1) and (2)]. It is pointed out that the interaction tendency is strongest when the surfactant is charged and the polymer is a polyion bearing charges of opposite sign to that of the surfactant. For example, if one "titrates" a polycation with an anionic surfactant, one obtains indications of "cooperative" binding of the surfac- tant, with formation of surfactant clusters, at very low levels of added surfactant. Evi- dence of the latter comes from enhanced solubilization of oil-soluble dyes (3) and of various fiuorescing agents (4). Furthermore, evidence of structural changes accompa- nying formation of the "complexes" has been obtained from such measurements as self-diffusion (5), small angle neutron scattering (6), and viscosity (6). Changes in the 19
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)