DYES AND SURFACTANTS FOR HAIR 11 Table V Change in Hunter b-Value of Untreated Blended Gray Hair Dyed With 2 or 4 After Six Shampooings pH of the Compound Concentration Dye medium Dye medium •Ab•* 2 55 mM 5 4 55 mM 5 2 55 mM 10 4 55 mM 10 2 24 mM 10 4 24 mM 10 2 55 mM 5 4 55 mM 5 4 55 mM 5 20% Ethanol 1.3 20% Ethanol 0. ! 20% Ethanol 2.3 20% Ethanol 1.5 60% Ethanol 0.9 60% Ethanol 1.5 20% Ethanol, 1.5 then pH 10 0.4 20% Ethanol, 0.0 then pH 10 (Ammonia gas, 18 h). * Ab -- b (initial) - b (after six shampooings). exposing the fibers to a dye solution at pH 5 and by a subsequent treatment of the hair at pH 10. Under these conditions, the isothiuronium-containing dye is more durable than its unreactive analog, 2. A further increase in the durability of 4 was observed in an experiment that involved treating a hair tress with a solution of 4 at pH 5, and then exposing it to ammonia vapors for 18 hours. In this case, there was no change in the b-value after six shampooings. Compounds 2 and 4 were also applied to hair in combination with reducing and oxi- dizing agents. The procedures involved (a) application of dyes after the reducing step and prior to oxidation, (b) simultaneous treatment of hair with a reducer and a dye followed by oxidation with H202, and (c) reduction followed by simultaneous oxidation and dyeing (Table VI). In all cases the isothiuronium-containing dye proved to be more substantive, the effect being the least evident when the dye is applied in the oxidation step. MICROSCOPIC ANALYSIS OF FIBER CROSS SECTIONS Distribution of the dyes in piedmont hair was assessed by examining micrographs of the Table VI Change in Hunter b-Value of Reduced Blended Gray Hair Dyed With 2 or 4 After Six Shampooings pH of the Compound Pretreatment Dye medium Dye medium Post-treatment •Ab•* 2 Thioglycolate 5.0 Solution Peroxide 1.9 4 Thioglycolate 5.0 Solution Peroxide 0.2 2 None 9.2 Thioglycolate Peroxide 0.9 4 None 9.2 Thioglycolate Peroxide 0.2 2 Thioglycolate 3.4 Peroxide None 1. ! 4 Thioglycolate 3.4 Peroxide None 0.8 * Ab = b (initial) - b (after six shampooings).
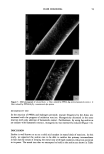
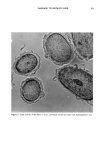
12 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS fiber cross sections after 30-min treatments. At pH 5, the isothiuronium-containing dye 4 does not penetrate throughout the fibers, but deposits primarily in the cuticle and the outer layers of the cortex (Figure 3). The treatments at pH 10 (not shown), for the partially or completely hydrolyzed isothiuronium moiety, led to uniform distributions of the dye throughout the fiber. Under the same conditions, at both pH 5 and 10, compound 2 readily diffuses and forms uniform deposits throughout the fiber (Fig- ure 4). DISCUSSION In order to analyze the interaction and substantivity of the isothiuronium-containing dyes and surfactants to hair, the following factors have to be taken under consideration: © Diffusion and entrapment of dyes and surfactants in the fiber structure. Contribution of the electrostatic, Van der Waals, and hydrogen-bonding forces to the binding of dyes and surfactants to the hair. © Formation of covalent bonds between the isothiuronium or thiol groups of a dye or a surfactant, and disulfide or thiol groups of the keratin fiber. © Entrapment of the absorbed dyes and surfactants by the change in their solubility or dimensions (steric effects) through the hydrolysis of the isothiuronium group or for- mation of dimeric products by oxidation of the thiol functionality. Based on its washfastness characteristics at pH 5 and 10, and the distribution profiles in fiber cross sections, the absorption and desorption of dye 2 follows a pattern similar to that observed for semipermanent, nonionic dyestuffs such as N-(2-hydroxyethyl)-2- nitro-p-phenylenediamine, studied quantitatively by Han et al. (19). The process of absorption of this dye was shown to be primarily diffusion-controlled. The dye uptakes were higher at low pH, which was ascribed to ionic binding of the protonated, and thus positively charged, dye molecules to negatively charged hair protein. However, absorp- Figure 3. Micrographs of piedmont hair treated with compound 4 at pH 5 (magnification 312 X ).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)