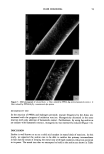
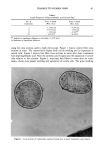
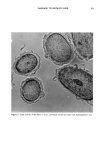
DYES AND SURFACTANTS FOR HAIR 15 previously reported in the literature, but it is thought to be possible because of the strong nucleophilic character of the thiol group. This results in attachment of the iso- thiuronium group to the keratin protein and the insolubilization of the dye in the fiber. Insoluble products can also be formed as a result of the dimerization of the thiol-substi- tuted dye. Covalent attachment of the dye to keratin can occur during the oxidation step (reaction 2). Also, oxidation of the dye to the sulfonic acid derivative, and the well known reactions of reduced keratin, are included in the scheme as viable processes (reactions 4-6). The relation between the surfactant structure, its mechanism of hair ad(b)sorption, and conditioning was investigated by Finkelstein et al. (20). They found that the sorption of the octadecyltrimethyl ammonium iodide proceeded much faster, and produced a more pronounced conditioning effect, than the dodecyltrimethylammonium chloride. In ad- dition to this, they noted that the desorption, observed after long time intervals in the adsorption experiments, was evident only in the case of a longer-chain quat. As an explanation, it was suggested that while dodecyttrimethyl ammonium chloride pene- trates deeply into the fiber structure and thus does not modify the fiber surface, the octadecyl derivative can rearrange from its initial distribution into surface hemimi- celles. These desorption and surface rearrangement processes, leading to a conditioning effect, were demonstrated for dodecyltrimethylammonium chloride-treated hair by sub- jecting it to prolonged heating in the moist state at 56øC. The data presented in this paper indicate that 0.5-hour exposure of hair to unreactive surfactants such as stearalkonium chloride and hydroxyl- or bromide-containing quats results in their penetration into the fiber bulk and the formation of surface deposits. This is supported by both the quantitative absorption data as well as the observed reduction in combing work. Shampooing should remove some of the surface surfactant deposits and result in a significant increase in combing work. This indeed occurs for the fibers treated with the surfactant solutions for very short periods of time during which only adsorption can take place. In the case of 0.5-hour treatments, the conditioning effect is still evident after four and even eight shampooings, suggesting that, similar to the effect described by Finkelstein et al. (20), desorption and surface rearrangement processes after shampooing may take place. It should also be mentioned that the ad(b)sorbed cationic surfactants can possibly interact with anionic detergents present in the shampoo (21). The precipitation of the cationic surfactant-anionic surfactant com- plexes on the fiber surface can potentially contribute to the observed conditioning ef- fects. The durability of the unhydrolyzed isothiuronium-containing surfactants on unmodi- fled hair might be attributed to both electrostatic interactions, increased by the pres- ence of the additional positively charged groups and Van der Waals forces. The adsorp- tion and desorption data, which showed relatively small rates and extents of deposition of the isothiuronium-containing quat into hair, suggest that this compound does not readily penetrate the bulk of the hair and possibly remains confined to the outermost layers of the fiber structure. The increased size of the isothiuronium-containing surfac- tant molecules might also contribute to such a distribution of the quat in the fiber. The data presented in Table III suggest that thiol/disulfide exchange reaction (Scheme IV) might be an important factor contributing to the high washfastness of the treat- ments performed with both single and double isothiuronium quats at pH 10.7 on
16 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS untreated hair. On the other hand, the interaction between the unhydrolyzed isothi- uronium surfactant and reduced hair, followed by oxidation with H202, might involve the reactions presented in Scheme V (reactions 1-3). It should be noted, however, that the surfactant thioIs or dimers are water-soluble and cationic, due to the presence of quaternary ammonium groups. Thus, electrostatic binding, rather than insolubiliza- tion, in addition to covalent binding, could be responsible for the observed increased durability of these materials. Finally, it should be pointed out that the bromide-containing surfactants can also react with the thiol groups of reduced hair by forming sulfides (Scheme V, reaction 7). The data in Tables III and IV, however, indicate very high combing work after the treat- ment, with subsequent reduction in combing forces as a result of shampooings. Al- though this effect is observed for all surfactants at pH 10.7, it is much more dramatic in the case of the bromide-containing surfactant applied to reduced hair. Also, the combing work after eight shampooings was high and comparable to the untreated con- trol. All these data seem to suggest either a complete lack of interaction of bromide quats with reduced hair or, conversely, very efficient absorption and binding of the quats in the bulk of the fiber, with little modification of the fiber surface. This last point is strengthened by the results of hair treatments with bromide-containing quats at higher concentrations (not reported in this paper) that demonstrate durable condi- tioning of reduced hair. CONCLUSIONS It has been shown that the substitution of either a semipermanent dye or a cationic surfactant with an isothiuronium group increases its substantivity to hair. Enhanced electrostatic interactions, entrapment as a result of dimerization or change in solubility, or covalent binding through the formation of disulfide bonds with the keratin could be possible mechanisms of this phenomenon. Since after long treatment times unreactive cationic surfactants also show high substantivity to hair, and are not readily removed by shampooing, the enhanced conditioning effects in the case of isothiuronium derivatives was ascribed to a specific distribution of the surfactant with its maximum concentration close to the fiber surface. REFERENCES (1) W. Patterson, W. Geiger, L. Mizell, and M. Harris, Role of cysteine in the structure of the fibrous protein, wool,.]. Res. Nat. Bur. Stand., 27, 839 (1941). (2) K. Hall and L. Wolfram, Application of the theory of hydrophobic bonds to hair treatments,.]. Soc. Cosmet. Chem., 28, 231 (1977). (3) D. E. Deem and M. M. Rieger, Mechanical hysteresis of chemically modified hair, .]. Soc. Cosmet. Chem., 19, 395 (1968). (4) JP 62/205-014-A, assigned to Kao Corp. (5) A. Shansky, Hair straightening through silylation: A preliminary investigation, Cosmetic Technology, 32 (March 1982). (6) EP 0159628, assigned to Revlon Inc. (7) L. J. Wolfram, Wool modification by deposition of reactive polymers, Appl. Polym. Symp., 18, 523 (1971). (8) S. M. Murkinshaw and D. M. Lewis, "Reactive Dyes for Wool," in Proceedings of the International
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)