52 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 4O 2O , 210 I i 0 40 60 Hamamelis Extract Content (wt%) Figure 6. The effect of hamamelis extract content for pre-treatment on coloring. The hair tresses were immersed in the hamamelis extracts which were prepared in various concentrations. They were colored in 0.1% PPDA solution-suspended 0.2 g Ag-zeolite containing 10 wt% silver. ELUTION OF SILVER IONS IN SOLUTION FROM ZEOLITE The effect of pH between 7.5 and 11.0 on the silver ion elution from zeolite was investigated. The determination of silver concentration was measured after 30-sec incu- bation. It was shown that the higher the pH was, the more easily silver ions eluted (Figure 9). The zeolite skeleton destruction that was known to occur in strong alkali solution was not recognized under these conditions because the steep increase of silver ion concentration was not observed. The silver ion elution is considered to be caused by the ion exchange reaction with the cation of the appropriate size in the solution. To make sure of that, the relationship between eluting silver ion concentration and sodium concentration in the solvent was investigated. Sodium ion concentration was adjusted to 0-150 mM by dilution of the Carmody buffer with distilled water. The elution of silver ions increased according to increase of sodium ion concentration, as was expected (Figure 10). In order to increase the amount of the eluting silver ions for effective coloring, the relationship with Ag-zeolite content was investigated. The eluting behavior of silver ions was measured under conditions of various contents of Ag-zeolite from 0 to 1.0 g in 4 ml Carmody buffer. Below 0.1 g, the eluting silver ion concentration increased smoothly, but it decreased gradually above this (Figure 11). This is considered to be because the viscosity of the solution increased. It shows that there is an optimum con- tent of Ag-zeolite for effective coloring. RATE OF PPDA OXIDATION REACTION BY SILVER ION SOLUTION The time course of the absorption at 470 nm was measured. In Figure 12, the effect of the silver concentration on the reaction is shown. It shows that the increase of silver concentration slowed down the reaction rate and increased the amount of the final product. In the case of PPDA concentration, the reaction rate and the amount of products were proportional to it (Figure 13).
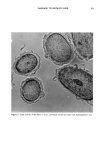
HAIR COLORING 5 3 0%,, ,:.-, ß i ' 7D Figure 7. SEM photograph of colored hairs. a: Hair colored by PPDA-Ag-zeolite-hamamelis extract. b: Hair colored by PPDA-H202, conventional dye system. MUTAGENICITY TEST In the reaction of PPDA and hydrogen peroxide, mutant frequency by the Ames test increased with the progress of oxidation reaction. Mutagenicity decreased in the same reaction with only addition of hamamelis extract. Furthermore, by using Ag-zeolite as an oxidant with hamamelis extract, mutagenicity was extensively reduced (Figure 14). DISCUSSION Zeolite is well known to act as a solid acid catalyst in many kinds of reactions. In this study, we expected the zeolite case to be able to oxidize the primary intermediates in hair dyes by means of keeping the metal ions with high oxidation-reduction potential in its pores. The metal ions that we attempted to hold in the zeolite are shown in Table
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)