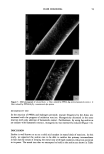
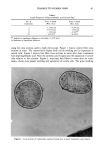
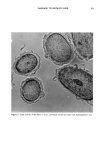
DYES AND SURFACTANTS FOR HAIR 13 Figure 4. Micrographs of piedmont hair treated with compound 2 at pH 5 (magnification 312 x ). tion-desorption experiments showed no sorption hysteresis, indicating a lack of a strong interaction between the dye molecules and the fiber. The absorption and desorption of dye 4 was expected to be strongly pH-dependent. At pH 5, the molecule possesses a cationic character and should bind to hair as a result of attractive electrostatic interactions. This might explain the relatively high durability of dyeouts produced by the use of this compound as compared to those obtained with the nonionic, or weakly cationic, compound 2. In addition to this, the analysis of the cross sections of the fibers treated with 4 at pH 5 showed nonuniform distribution of the dye throughout the fiber, with the highest concentration of the dye deposits close to the fiber surface (Figure 3). This suggests a strong interaction between the dye molecules and the fiber protein. At pH 10, the hydrolyzed molecules of 4 lose the ability to electrostatically interact with the fiber. This is supported by the micrographs of the fiber cross sections showing uniform dye distributions, similar to those obtained for the nonionic compound 2. On the other hand, the presence of reactive thiol groups in the hydrolyzed 4 creates the possibility of a thiol/disulfide exchange reaction and covalent binding of the dye to the hair as shown in Scheme IV. Such a reaction could contribute to the high substantivity of the dye to the hair that was observed with treatments Scheme IV. R--$H + K--S--S--K , R--$--$--K + HNCH2 CH2- uhere R = or CH.•(CH2) •4CH2•N--CH2CH2-
14 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS performed at low pH followed by a prolonged exposure of the fibers to a high-pH medium. Similar reactions were reported to occur between certain azo dyes containing disulfide links and the model compounds cysteine and thioglycolic acid, and wool ker- atin (11). However, an alternative explanation of the observed high substantivity of the hydrolyzed isothiuronium dyes could be their immobilization in the fiber structure as a result of the change in their water solubility. Such an explanation was proposed to account for the washfastness of a number of isothiuronium-containing dyes in wool staining experiments (9,10). The relatively high substantivity of the isothiuronium-containing dye to reduced hair, observed for the treatments involving the application of dyes after the reducing step and prior to oxidation, and in simultaneous treatments of hair with a reducer and a dye followed by oxidation with H202, could be explained by the reactions presented in Scheme V. The reaction [1] between the thiol and the isothiuronium group was not Scheme V. + ,•NH2 X-- (1) R•H 2CH •--S• •/' + K--SH •NH2 uahydrolyzed •NH2 X , R•H=CH=--SH + K--S• • •NH 2 (2) R--CH2CH2--SH + K--SH hydrolyzed H=02 • R•H2CH2--S--S--K (3) 2 R•H2CH2--SH H202 • R•H=CH=--S--S•H=CH2--R (5) K--S- H=O= • K--SO 3- (7) R•H2CH=--Br + K--SH • R•H=CH•S--K vhere R = or CH3 X- +1 CHa(CH2) 14CH2--N-- I CH3 and K = Keratin Protein
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)