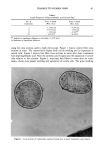
DYES AND SURFACTANTS FOR HAIR 7 cortex. The surfactant containing the hydroxyl group deposits on/in the fibers at a slightly faster rate than the bromide derivative at both pH 5.3 and 10.7. The rates and the extents of the deposition of the isothiuronium-containing surfactant were much smaller, especially at low pH. Table I presents the results of sorption experiments obtained for reduced hair. The fibers were treated with ammonium thioglycolate (6% adjusted to pH 9) for 10 min at room temperature and rinsed with deionized water for 1 min, prior to deposition. The reduced hair readily absorbs the hydroxyl and bromide-containing surfactants, and within the first half hour, the solutions are almost completely depleted. Similar to the results obtained for untreated hair, the isothiuronium-containing surfactant deposited in relatively small quantities in/on reduced fibers. The size of the isothiuronium deriva- tive, its charge characteristics, and reactivity towards the thiol groups might contribute to its slow rate of deposition in hair. The extraction data presented in Table II show that only a small fraction of the ad(b)sorbed hydroxyl- (16.0% and 11.4% for hair treated at pH 10.7 and 5.3, respec- tively) and bromide- (20.7% and 21.1% for hair treated at pH 10.7 and 5.3, respec- tively) containing surfactants can be extracted from hair. Extraction of hair treated with the isothiuronium-containing surfactant results in the removal of significantly larger quantities of the deposited material (41.3% and 80.7% for hair treated at pH 10.7 and 5.3, respectively). This result seems to indicate, in agreement with the sorption data, 40 pH 10.7 • •_ :30 __-J-- •J • 25 • -- 1 0 • 4 8 • 1'0 1• 1'4 1 1'8 •0 2• 24 '6 Time (hours) [] alcohol (11) + bromide (12) • isothiuronium (13) Figure 2. Amount of surfactant ab(d)sorbed as a function of time for unmodified hair at pH 10.7.
8 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table I Amount of Surfactant Ad(b)sorbed by Reduced Hair Treated at pH 10.7 Amount ad(b)sorbed Compound Time (hours) (mg/g) 11 0.5 33.78 12 0.5 37.73 13 0.5 19.26 that the deposited isothiuronium-containing quaternary salt does not penetrate deeply into the fiber structure and that most of it does not form covalent bonds with keratin. CONDITIONING EFFECTS The ability of 1ong-alkyl-chain quaternary ammonium salts to improve dry and wet combability of hair is well known and has been previously described in the literature (17). Low-molecular-weight quat treatments such as cream rinses are, however, not durable, since the adsorbed surfactant can sometimes be removed by shampooing. We expected that the incorporation of a reactive moiety into the surfactant structure might result in covalent binding of a surfactant to keratin, and therefore improve the dura- bility of the conditioning effect. Wet combing measurements, obtained following the treatments with a series of com- pounds 5-10, and after four and eight shampooings, were used to assess the condi- tioning effect. The treatments were performed at pH 5.3 (unhydrolyzed isothiuronium salt) and pH 10.7 (partially hydrolyzed isothiuronium salt). Stearalkonium chloride, a widely used cationic surfactant in cream rinse formulations, was also tested as a control. Table III shows the wet combing data obtained for the experiments performed on un- modified hair. For the control (untreated tresses) the wet combing work was found to increase after four and eight shampooings. This phenomenon is not well understood, although it may be caused by the progressive damage to the hair cuticle caused by shampooing, combing, and handling in the wet state (18). The treatment with the unreactive surfactant, stearalkonium chloride, reduced the wet combing work both at pH 5.3 and 10.7. The surfactant is not completely removed from the fibers after eight shampooings as evidenced by the reduced values of combing work as compared to the untreated control. Note that the value of the combing work obtained immediately after Table II Quantity of Surfactant Removed From Unmodified Hair Treated at pH 5.3 or 10.7 Amount desorbed (mg/g) pH of the Compound Treatment After 2 hours After 72 hours 11 5.3 2.76 10.7 2.54 12 5.3 5.84 10.7 5.61 13 5.3 5.19 10.7 3.98 3.72 (11.4%) 5.48 (16.0%) 6.86 (21.1%) 7.05 (20.7%) 6.26 (80.7%) 6.57 (41.3%)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)