BRING OVER 1,000+ HOURS OF COSMETIC SCIENCE CONTENT TO YOUR HOME. On the SCC Media Library and Resource Center, you’ll find everything from FREE articles in the Journal of Cosmetic Science and SCC sponsored webinars to LIVE recordings of scientific sessions presented at the Society’s Annual Meeting. Start exploring today! www.scconline.org/library
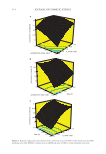
362 Address all correspondence to Melisa Lalikoglu at melisad@iuc.edu.tr. J. Cosmet. Sci., 72, 362–378 (July/August 2021) The Synthesis of Benzyl Acetate as a Fragrance and Flavoring Agent with Green Solvents MELISA LALIKOGLU AND EROL İNCE Istanbul University-Cerrahpasa, ¸ Engineering Faculty, Chemical Engineering Department, 34320, Istanbul/TURKEY Accepted for publication May 2, 2021. Synopsis Benzyl acetate, which has a crucial role in the industry as a flavor and fragrance component, is important for human health to be obtained with a green and clean process. For this purpose, the esterification reac- tion of acetic acid (AA) and benzyl alcohol (BA) was investigated using five different ionic liquids (ILs) as catalysts. 1-Ethyl-3-methylimidazolium hydrogen sulfate, [EMIM] [HSO 4 ], 1-Ethyl-3-methylimidazolium tetrafluoroborate [EMIM] [BF 4 ], 1-methyl-3-octylimidazolium tetrafluoroborate [OMIM] [BF 4 ], 1-ethyl-3- methylimidazolium bis [(trifluoromethyl)sulfonyl] imide [EMIM] [NTf 2 ], and 1,3-diethylimidazolium bis [(trifluoromethyl)sulfonyl] imide [DEIM] [NTf 2 ] were used as catalysts. The best catalytic performance was obtained with 1-Ethyl-3-methylimidazolium hydrogen sulfate, [EMIM] [HSO 4 ]. The influence of different anions and cations in the IL’s structure, the reaction conditions such as initial acid/alcohol ratio, the catalyst amount, the reaction temperature, and the time on conversion were observed. The Box–Behnken experimen- tal design of response surface methodology was applied to estimate the relationship between acid conversion (%) and reaction parameters. According to the model, in all esterification experiments carried out at 110°C, the optimum conditions for maximum conversion were AA:BA molar ratio of 1:1, IL molar ratio of 0.66, and reaction time 4 hours. Under these conditions, 90.34% acid conversion was achieved. [EMIM] [HSO 4 ] can be used for up to three cycles with minimal loss in activity. INTRODUCTION Organic esters are valuable products in the chemical industry. One of the short-chain carboxylic acid esters, benzyl acetate, is used as a flavor and fragrance component in the food and cosmetics industry. The Council of Europe approved the use of benzyl acetate in foodstuffs in 2000 and included in the list of substances granted-A. In addition, it is considered safe as a flavor ingredient by the Flavor and Extract Manufacturers Association (FEMA). Although benzyl acetate is found naturally in cloves, chamomile, jasmine, and hyacinth, extracts obtained from these plants are not sufficient to meet the worldwide usage volume (about 10,000 tons per year). Therefore, it is obtained by many different chemical methods. Most commonly used method is the esterification reaction of acetic acid (AA) and benzyl alcohol (BA) (1–2).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)