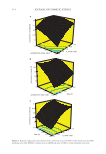
375 SYNTHESIS OF BENZYL ACETATE highest conversion, was investigated in benzyl acetate esterification reaction. The exper- iments were carried out with AA:BA:[EMIM] [HSO 4 ] molar ratio of 1:1:075 at 110° C for 8 hours. At the end of the reaction, the upper phase and the lower phase (IL and water) were separated by simply decantation. The IL was dried under vacuum (0.01 Torr) at 100° C (overnight). The reusability of the IL was investigated by repeating the same experimental procedure five times for the recycled IL. Figure 7 represents that the cata- lytic activity of [EMIM] [HSO 4 ] did not change until the third run, but began to decrease slightly with the next use. CONCLUSIONS In this study, the obtaining of benzyl acetate, which gives artificial jasmine scent and apple, banana flavors to various cosmetics and personal care products like perfumes, lotions, hair creams, and perfumes, by esterification method was investigated. For this purpose, five different ILs were used as a catalyst for the esterification reaction of AA with BA. The results showed that the highest acid conversion was obtained in the ester- ification reaction, in which [EMIM] [HS0 4 ] was used as a catalyst. The response surface methodology based on the Box–Behnken design for optimizing the reaction parameters was applied using this IL. The optimum conditions were AA:BA molar ratio of 1:1, IL molar ratio of 0.66, and reaction time of 4 h. Under these conditions, 90.34% acid conversion was achieved. ANOVA was performed to test the suitability of the proposed second-degree model in order to determine the effect levels of the independent variables affecting the acid conversion. According to the statistical results obtained, it was seen that the most crucial factor affecting the reaction was the amount of IL. As a result, the IL used in this study proved to be an excellent catalyst for the esterification of AA and BA. This study has been a green approach obtaining the ester with high efficiency, and easy separation and recovery of the IL used as a catalyst from the product. In future research, it is important to study that different esters, which are frequently used in personal care products, can also be obtained using this method. ACKNOWLEDGMENTS This study was funded by Istanbul University with Project Number 34009. REFERENCES (1) D. McGinty, D. Vitale, C. S. Letizia, and A. M. Api, Fragrance material review on benzyl acetate, Food Chem Toxicol., 50, 363–384 (2012). (2) R, Lamba, S. Kumar, and S. Sarkar, Esterification of decanoic acid with methanol using Amberlyst 15: reaction kinetics, Chem Eng Commun., 205, 281–294 (2018). (3) J. Lilja, J. Aumo, T. Salmi, D. Y. Murzin, P. M. Arvela, M. Sundell, K. Ekman, R. Peltonen, and H. Vainio, Kinetics of esterification of propanoic acid with methanol over a fibrous polymer-supported sulphonic acid catalyst, Appl Catal A-Gen., 228, 253–267 (2002). (4) H. J. Bart, J. ReIdetschlager, K. Schatkaj, and A. Lehmann, Kinetics of esterification of levulinic acid with n-butanol by homogeneous catalysis, Ind Eng Chem Res., 33, 21–25 (1994) (5) J. Lilja, D. Y. Murzin, T. Salmi, J. Aumo, P. Maki-Arvela, and M. Sundell, Esterification of different acids over heterogeneous and homogeneous catalysts and correlation with the Taft equation, J Mol Catal-A Chem, 182, 555–563 (2002).
376 JOURNAL OF COSMETIC SCIENCE (6) R. A. Troupe and K. A. Kobe, Kinetics of methanol-lactic acid reaction, Ind Eng Chem Res., 42, 1403–1409 (1950). (7) R. A. Troupe, K. A. Kobe, Kinetics of ethyl alcohol lactic acid reaction, Ind Eng Chem Res., 49, 847–855 (1957). (8) Y. Liu, E. Lotero, and J. G. Goodwin, Effect of carbon chain length on esterification of carboxylic acids with methanol using acid catalysis, J Catal., 243, 221–228 (2006). (9) F. Xu, H. Chen, H. Zhang, X. Zhou, and G. Cheng, Protophilic amide ionic liquid assisted esterifica- tion and catalysis mechanism, J Mol Catal-A Chem., 307, 9–12 (2009). (10) Y. Tsai, H. Lin, and M. Lee, Kinetics behavior of esterification of acetic acid with methanol over amber- lyst 36, Chem Eng J., 171, 1367–1372 (2011). (11) P. Delgado, M. T. Sanz, and S. Beltran, Kinetic study for esterification of lactic acid with ethanol and hydrolysis of ethyl lactate using an ion-exchange resin catalyst, Chem Eng J., 126, 111–118 (2007). (12) Y. Seo and W. H. Hong, Kinetics of esterification of lactic acid with methanol in the presence of cation exchange resin using a pseudo-homogeneous model, J Chem Eng Jpn., 33, 128–133 (2000). (13) G. D. Yadav and H. B. Kulkarni, Ion-exchange resin catalysis in the synthesis of isopropyl lactate, React Funct Polym., 44, 153–155 (2000). (14) A. Engin, H. Haluk, and K. Gurkan, Production of lactic acid esters catalyzed by heteropoly acid supported over ion-exchange resins, Green Chem., 5, 460–466 (2003). (15) Y. Zhang, L. Ma, and J. Yang, Kinetics of esterification of lactic acid with ethanol catalyzed by cation- exchange resins, React Funct Polym., 61, 101–114 (2004). (16) J. Gangadwala, S. Mankar, S. Mahajani, A. Kienle, and E. Stein, Esterification of acetic acid with butanol in the presence of ion-exchange resins as catalysts, Ind Eng Chem Res., 42, 2146–2155 (2003). (17) S. H. Ali and S. Q. Merchant, Kinetic study of Dowex 50 Wx8-catalyzed esterification and hydrolysis of benzyl acetate, Ind Eng Chem Res., 48, 2519–2532 (2009). (18) P. E. Jagadeeshbabu, K. Sandesh, and M. B. Saidutta, Kinetics of esterification of acetic acid with meth- anol in the presence of ion exchange resin catalyst, Ind Eng Chem Res., 50, 7155–7160 (2011). (19) M. Lee, J. Y. Chiu, and H. Lin, Kinetics of Catalytic Esterification of propionic acid and n-Butanol over Amberlyst 35, Ind Eng Chem Res., 41, 2882−2887 (2002). (20) V. V. Mali and M. P. Deosarkar, Esterification of acetic acid with butanol in the presence of solid acid catalyst, Intl J Adv Sci Eng Technol., 4, 245−248 (2016). (21) A. Mittal, S. Nair, and K. Deshmukh, The kinetic comparison study of catalytic esterification of butyric acid and ethanol over Amberlyst 15 and Indion-190 resins, Intl J Innovat Res Sci Eng Technol., 4, 5860−5867 (2015). (22) T. Suman, S. Srinivas, and S. M. Mahajani, Entrainer based reactive distillation for esterification of eth- ylene glycol with acetic acid, Ind Eng Chem Res., 48, 9461–9470 (2009). (23) A. Chinnappan and H. Kim, Environmentally benign catalyst: Synthesis, characterization, and proper- ties of pyridinium dicationic molten salts (ionic liquids) and use of application in esterification, Chem Eng J., 187, 283–288 (2012). (24) P. Kubisa, Application of ionic liquids as solvents for polymerization processes. Prog Polym Sci., 29, 3–12 (2004). (25) J. Lu, F. Yan, and J. Texter, Advanced applications of ionic liquids in polymer science, Prog Polym Sci., 34, 431–448 (2009). (26) S. Singhal, S. Agarwal, M. Sing, S. Rana, S. Arora, and N. Singhal, Ionic liquids: green catalysts for alkene-isoalkane alkylation, J Mol Liq., 285, 299–313 (2019). (27) C. P. Huang, Z. C. Liu, C. M. Xu, B. H. Chen, and Y. F. Liu, Effects of additives on the properties of chloroaluminate ionic liquids catalyst for alkylation of isobutane and butene, App Catal A-Gen., 277, 41–43 (2004). (28) Z. Zhu, Y. Ri, M. Li, H. Jia, Y. Wang, and Y, Wang, Extractive distillation for ethanol dehydration using imidazolium-based ionic liquids as solvents, Chem Eng Process., 109, 190–198 (2016). (29) G. Yu, C. Dai, L. Wu, and Z. Lei, Natural gas dehydration with ionic liquids, Energy Fuels, 31, 1429– 1439 (2017). (30) K. R. Seddon and A. Stark, Selective catalytic oxidation of benzyl alcohol and alkylbenzenes in ionic liquids, Green Chem., 4, 119–123 (2002). (31) I. F. Mena, E. Diaz, C. Pérez-Farías, S. Stolte, I. Moreno-Andrade, J. J. Rodriguez, and A. F. Mohedano, Catalytic wet peroxide oxidation of imidazolium-based ionic liquids: catalyst stability and biodegrad- ability enhancement, Chem Eng J., 376, Article ID: 120431 (2019).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)