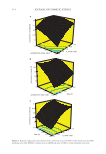
373 SYNTHESIS OF BENZYL ACETATE of 0.9617 and R2 adj value was found 0.9124. These two values were close to each other shows that the conversion of acid (%) estimated by the obtained model equation was quite adjacent to the conversion of acid (%) obtained by experimental data. “Adeq Precision” measures the signal-to-noise ratio. A ratio greater than 4 is desirable. The ratio of the model found as 16.556 indicated an adequate signal. This model can be used to navigate the design space. These statistical data showed that the quadratic model obtained can be used to predict acid conversion (%) within the range of the variables studied. The three dimensional and contour plots of the variables on the conversion of acid (%) are shown in Figure 8A, B, and C. These graphs show the mutual effect of the other two parameters by keeping one of the parameters constant (61). Figure 8A represents the interaction between acid/alcohol molar ratio and IL molar ratio. Conversion of acid (%) decreased with acid/alcohol mole ratio and increased with IL mole ratio. More than one of the reactants causes the balance to shift toward the products. However the increase in the acid/alcohol mole ratio, the excess acid (%) in the medium did not increase the acid conversion (%). It can be explained by the dilution of alcohol and IL with excess acid in the reaction medium, as in previous studies (63,67,73). The interaction between acid/alcohol mole ratio and time is shown in Figure 8B. It is clear that the conversion of acid (%) increased with increasing the reaction time and decreased with the acid/alcohol mole ratio. Figure 8C shows the interaction between time and IL molar ratio. The plot revealed that the conversion of acid (%) decreased slightly with time, and at the point where the IL mole ratio is maximum, the conversion of acid (%) is the highest. This might be due to active sites in the reaction medium increase with the amount of IL (74). As a result, the IL molar ratio is a more important parameter than the reaction time for the esterification reaction also it can be seen from the ANOVA table. RECYCLE AND REUSE OF IL One of the most important factors in the ability of ILs to reduce the cost of the ester- ification process is the possibility to be reused. The recycling and reuse of the used IL will both reduce the production cost and its impact on the environment because it is not thrown into the environment (62). The reusability of [EMIM] [HSO 4 ], which had the 0 10 20 30 40 50 60 70 80 90 100 1 cycle 2 cycle 3 cycle 4 cycle 5 cycle Figure 7. Performance of recycled [EMIM] [HSO 4 ] catalyst (Reaction Conditions:AA: BA: [EMIM] [HSO 4 ] = 1:1: 0.75, T = 110°C, t = 8 h). Conversion of acid (%)
374 JOURNAL OF COSMETIC SCIENCE A B C 1.00 1.25 1.50 1.75 2.00 0.25 0.38 0.50 0.63 0.75 47 57.25 67.5 77.75 88 acid/alcohol molar ratio IL molar ratio 1.00 1.25 1.50 1.75 2.00 4.00 5.00 6.00 7.00 8.00 65 70.75 76.5 82.25 88 acid/alcohol molar ratio time (h) 0.25 0.38 0.50 0.63 0.75 4.00 5.00 6.00 7.00 8.00 61 68.25 75.5 82.75 90 IL molar ratio time (h) Figure 8. Response surface plots and contours of the conversion of acid. (A) Effect of the molar ratio of AA:BA and molar ratio of IL, (B) Effect of molar ratio of AA:BA and time, (C) Effect of time and molar ratio of IL. %) Convesonofacd %) Convesonofacd %) onesonofacd C
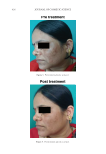
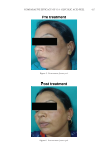
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)