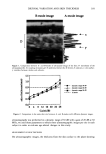
HAIR PHOTOPROTECTION BY DYES 385 1.6 1.2 Dyed with Red Permanent Color 216 h Irradiation Piedmont Hair • • 200 300 400 500 600 700 800 900 Wavelength (nm) Figure 5. Fading of hair color upon photoirradiation. Absorption spectra calculated from the reflectance data for Piedmont hair before dyeing. Hair dyed with the red shade of a permanent ha • -lor and dyed tress after 216 hours (nine days) of photo-exposure in the solar simulator. intensity at 510 cm -• due to photo-exposure was used to calculate hair damage (see Experimental section). The photoprotection afforded by the three semipermanent dye products to the Piedmont hair is shown in Figure 8. The first, SPl(b•ue), is a bluish shade while the other two, SPl(red ) and SP2(r•d), are auburn (red). The data show that the two red shades produce similar photoprotection, while the blue shade is somewhat less efficacious. Absorption measurements (data not shown) reveal that SPl(b•u o deposited less color on hair, com- pared to the other two products. This may explain, at least in part, the observed difference in the efficacy of photoprotection. The dyes used in these products are mainly substituted benzenes, and the blue shade contains some aminoanthraquinones, in addi- tion. Besides absorption in the visible wavelength region, which is responsible for the color, these dyes also absorb UV light. This combination results in the observed pro- tective effect. It is likely that since the red dyes absorb, and thereby attenuate, the more energetic part of the spectrum, compared to the blue dyes, they may be intrinsically somewhat more protective. It should be mentioned that unlike the products based on the oxidation dyes, the semipermanent coloring products only deposit the color without affecting the natural pigment. Therefore, as has been discussed above, their efficacy in protecting hair against photodamage should not depend on the natural color of the hair. In a recent paper, Hoting and Zimmerman (5) have also concluded that coloring of human hair with oxidation dyes protects against photodamage to proteins and the internal lipids. They suggest that oxidative dyeing alters the protein structure, thereby
386 JOURNAL OF COSMETIC SCIENCE 12 9 6 ß Undyed Natural Brown ß Dyed with Dark Brown Permanent Color ß Dyed with Dark Brown Demi-Permanent Color i i 0 3 6 9 Photoexposure (days) Figure 6. Hair damage resulting from photoirradiation of natural brown (pigmented) hair before and after dyeing with the brown shades of either a permanent or a demipermanent hair color. Notice that in contrast to the data shown in Figure 4, in this case the demipermanent color provides better photoprotection than the permanent color (see text for details). limiting the extent of damage that photochemically generated hydrogen peroxide can cause. Our data show that both the oxidation dyes and the direct dyes provide photo- protection. Since the latter are not likely to alter the protein structure, we feel that the hair dyes act as passive photofilters and reduce damage by attenuating the incident light. The interaction of light with a dye molecule can produce a wide variety of effects. The simplest case is the absorption of light, which promotes the dye molecule to the excited state, followed by a return to the ground state via radiative and non-radiative pathways. The net result is a rather innocuous photofiltering effect. Complications occur when the excited state of the molecule undergoes photochemical reactions. The intermediates in the reaction pathway and/or the final product may accentuate damage. In a complex mixture of dyes, such as those used in hair-dyeing products, the results are harder to
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)