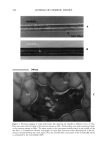
LIPOIC ACID STABILITY 455 room temperature (20 ø + 2øC). The flow rate was 1.8 ml/min, with UV detection at 325 nm for vitamin A and at 230 nm for vitamin E. The flow rate was 0.6 ml/min, with UV detection at 332 nm for lipoic acid. The volume of each injection was 20 pl. In these conditions, the vitamin A, vitamin E, and lipoic acid retention times were nine, three, and six minutes, respectively. Procedure. Solutions of the vitamins and lipoic acid were prepared on a weight basis with volumetric flasks to minimize solvent evaporation. Prior to injecting the solutions, the column was stabilized for at least 30 minutes with the mobile phase flowing through the system. Quantification was accomplished using an external standard method. Each solution was prepared in duplicate and was injected in triplicate. The relative standard deviation (RSD) was below 2.0%. Working standard solutions. Twenty milligrams of vitamin A were placed into a 50-ml volumetric flask, dissolved in 40 ml of isopropyl alcohol, shaken for about five minutes, and then diluted to volume with isopropyl alcohol. The standard preparation was obtained by diluting 4 ml of the vitamin A stock solution with mobile phase to yield a concentration of 0.016 mg/ml. Fifty milligrams of vitamin E were taken in a 50-ml volumetric flask, dissolved in 40 ml of isopropyl alcohol, shaken for about five minutes, and then diluted to volume with isopropyl alcohol. The standard preparation was obtained by diluting 4 ml of the vitamin E stock solution with mobile phase to yield a concentration of 0.04 mg/ml. Twenty-five milligrams of lipoic acid were taken in a 25-ml volumetric flask, dissolved in 20 ml of methanol, shaken for about five minutes, and then diluted to volume with methanol. The standard preparation was obtained by diluting 8 ml of this acid stock solution with mobile phase to yield a concentration of 0.08 mg/ml. Preparation ofo/w samples. Around 450 mg of cream were exactly weighed, placed into a 25-ml volumetric flask, taken to volume with methanol, and shaken for about five minutes for vitamin analysis. About 450 mg of cream were exactly weighed and placed into a 25-ml volumetric flask, taken to volume with mobile phase, and shaken for about five minutes for lipoic acid analysis. The solutions were passed through a 0.45-micron membrane filter before injection. Selectivity. Method selectivity was determined by degrading vitamins A and E and lipok acid as follows: Drugs were subjected to photochemical degradation (2 ml in an open A B C D 1• Figure 5. Vitamin A: (A) Standard. (B) Water degradation. (C) Photochemical degradation. (D) Acid degradation. (E) Oxidant degradation.
456 JOURNAL OF COSMETIC SCIENCE Figure 6. Vitamin A: Typical chromatogram of the o/w formulations. container exposed to daylight for 72 or 96 hours, not controlled esposure). Twenty milligrams of vitamin A were dissolved in 25 ml of isopropyl alcohol. Two milliliters of this solution were dissolved in 10 ml of water, 10 ml of HCI iN, and 10 ml of H202, 100 vol., and refluxed for at least 30 minutes. Degradation was monitored as a function of
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)