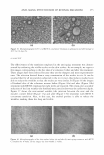
302 JOURNAL OF COSMETIC SCIENCE then converted to the L* and a* values of the CIE color model, using equations (1) and (2) presented in the Materials and Methods section. Topical treatments for eight weeks with dA (Figure Sa) resulted in a gradual improve ment in skin lightening compared to the vehicle-treated control (i.e., increase in 11.L* values higher values for dL* indicate the occurrence of lightening). HQ and TBP treatment also resulted in a gradual increase in skin lightening. Statistical analysis demonstrated that skin lightening at weeks 4, 6, and 8 in all treated groups was statistically different from the initial L value in each group (p 0.0001). The data in Figure Sa is also presented in a bar graph (Figure Sb) that includes the standard deviation (standard error, SE) per group. Standard deviation was relatively greater in the HQ- and TBP-treated groups at weeks 4, 6, and 8 than in the respective dA-treated groups. Histological sections of xenografts were obtained at the termination of treatment (i.e., week 8) and evaluated for percent melanin per epidermal area as described in Material and Methods. Treatment with all tyrosinase inhibitors resulted in a significant 12 ------------------- - 10 � 8 ai C: c f 6 ai � 4 a O 0 - Conlrol · V · cl,f\ ------- HO - --- 18P 2wb * 4wb 8wb 8wb 16 �------------------ 14 l 12 ai 10 C S 8 � ai ::::i 8 _J j 4 a 2 c::::J Con1rol IIIIIIl dA l!ail!llBIHO -TBP 2wk• 4wks 6wk• 8wb Figure 5. Topical treatment for eight weeks with dA resulted in a gradual increase in skin lightening compared to the vehicle-treated group. Human skin grafted onto mice (three mice per group) was treated with 5% test compounds, five days per week, for eight weeks. Treatment sites were assessed biweekly for the degree of pigmentation using the Charm View™ system. The Land a values for each treatment site were obtained by using Adobe Photoshop software and then were converted to the L* and a* values of the CIE color mode. The time course of change in L* values from baseline value for grafts treated with dA, HQ, TBP, and vehicle are presented as (a) a line graph and (b) a bar graph depicting the mean ± SE. * = Significant difference atp 0.0001 between the baseline value for each group and their values at four-, six-, and eight-week points.
EFFICACY AND SAFETY OF DEOXYARBUTIN 303 inhibition of the melanin present in the epidermis (Table III). The lightening of grafting-induced hyperpigmentation was visibly apparent (Figure 6a-f). In addition, two of three mice treated with HQ developed brown coloration of the hair around the grafts not observed in mice treated with the vehicle or dA (Figure 6g-i). H&E stained sections for grafts treated with dA and HQ for eight weeks demonstrated no signs of inflammation or abnormal morphology (data not shown). CLINICAL TRIAL RESULTS The effect of dA and HQ on the lightening of tanned skin was compared in a clinical trial as described in the Materials and Method section. After a seven-day regimen of tanning, areas of skin were left untreated or treated blindly with either dA or HQ, three times per week for five weeks. At the end of this period, the percent of tan remaining in the untreated site was 44.6%. In comparison, the percent of tan remaining in the sites treated with either dA or HQ was 37.3% (a significant increase in tanning loss over control) versus 5 J 6% (a sigoificaor decrease in tanning loss over control), respectively. These results suggest that dA accelerated the fading of UV-induced tan, whereas HQ impeded this response. DISCUSSION Toxicity of phenolic compounds can arise from three possible mechanisms: (a) suscep tibility of agents to extracellular auto-oxidation, (b) cellular permeability, and (c) in tracellular oxidation by tyrosinase and/or other metabolic enzymes (25-27). Deoxyar butin (dA), was shown to reversibly reduce tyrosinase activity and melanin content and demonstrated less cytotoxicity, as compared to HQ, in the three normal human skin cell types (i.e., melanocytes, keratinocytes, and fibroblasts). Studies have previously demonstrated that a major component in the toxicity of phenolic compounds is attributable to reactive oxygen species produced outside the cells (2 5 ). Specifically, hydroquinone auto-oxidation was shown to occur predominantly in the extracellular environment and to be a causative event for quinone-induced cytotoxicity (27). Thus, the lower cytotoxicity of dA compared to HQ may be due to its enhanced stability and reduced auto-oxidation. The enhanced stability could be attributed to the presence of an acetyl bond in its structure that allows stability in basic conditions. In addition, and in contrast to HQ, the OH group in the para position forming the acetyl bond in dA would result in conferring dA less susceptible to auto-oxidation. Table III Percent Melanin per Epidermal Area in Xenografts Treated for Eight Weeks with Vehicle or Various Tyrosinase Inhibirors Vehicle dA HQ TBP % Melanin/epidermal area 0.088 ± 0.07 0.061 ± 0.06 0.043 ± 0.05 0.015 ± 0.02
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)