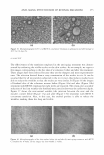
304 dA HQ 4-TBP JOURNAL OF COSMETIC SCIENCE Pretreatment \1-� · ·?"'':� ...._ -- !"'I- _,._.. 8 Wk treatment Figure 6. Grafting-induced hyperpigmentation is reduced by dA. Grafts described in Figure 5, at the onset of the treatment (a,c,e) and after eight weeks of treatment (b,d,f) with dA (a,b), HQ (c,d), or TBP (e,f), demonstrate the skin-lightening effect of each tyrosinase inhibitor. In addition, grafts treated for eight weeks with vehicle only (g) as opposed to dA (h) or HQ (i) exhibit darker versus lighter pigmentation, respectively. Note that in two of the three HQ-treated grafts a brown stain coloration adjacent to murine hair (arrows in i) is observed. The relative cellular permeability of phenols may also correlate with the effect of the compound on cellular viability, as well as its efficacy as its tyrosinase inhibitor (25). Relatively non-polar phenols, like dA and HQ, are more permeable and thus have the potential to be more toxic as compared to more polar phenols, like AR, that less readily enter the cell (25). Our present analysis demonstrates that AR is a relatively safe compound compared to dA and HQ. However, AR is either an ineffective tyrosinase inhibitor or promotes tyrosinase activity in light-versus-dark human melanocytes, re spectively. The ineffectiveness of AR as a tyrosinase inhibitory agent is in agreement with the findings of Curto et al. ( 15 ). However, an increase in pigmentation by AR has been previously reported in several studies (19,20). In contrast, several studies have demonstrated that AR is a tyrosinase inhibitor (17-19). However, these latter studies utilized high concentrations of AR in a culture system (17,19) and/or assessed tyrosinase inhibition using cell lysate rather than intact cells (17-19). This suggests that AR, although an effective inhibitor of the catalytic activity of isolated tyrosinase, is relatively
EFFICACY AND SAFETY OF DEOXY ARBUTIN 305 ineffective on intact melanocytes. The sugar residues in AR confer a significant shift in both size and polarity, e.g., the clog P of arbutin is -0.58 as compared to +0.56 for HQ, thus likely preventing it from passing through cellular membranes in sufficient con centration to inhibit tyrosinase within melanosomes. It is noteworthy to mention that, so far, the effective topical concentration of arbutin has not been formally evaluated and published (28). Once internalized, phenolic derivatives can act as substrates for tyrosinase. This requires electron donor groups (i.e., alkoxyl and hydroxyl groups) in the para position with respect to the OH group (5 ). HQ and its esters possess an electron donor group in that position. It has been demonstrated that HQ, an effective tyrosinase inhibitor (29), can be oxidized by tyrosinase to hydroxybenzoquinone and p-benzoquinone, thus generating reactive oxygen species (6,7 ,30). With dA and AR, the electron-donating ability of the para oxygen is reduced by the presence of a second oxygen ring, thus slowing or stopping the oxidation rate. Oxidation products can be produced from arbutin by mushroom tyrosinase (31). Whether dA can also be converted to quinones and generate reactive oxygen species, and at what rate, has yet to be determined. All tested tyrosinase inhibitors eventually demonstrated toxicity against keratinocytes and fibroblasts. This suggests that toxicity can arise from a non-tyrosinase-mediated mechanism. A peroxidase-mediated mechanism in the cytotoxic effects of melanogenic inhibitory agents, specifically phenolic compounds, has been demonstrated previously (32). Phenolic compounds can also serve as substrate and inhibitors for peroxidase (32-34). For example, HQ serves as a good substrate for peroxidase (35). Melanocytes, fibroblasts, and keratinocytes are all equipped with peroxidases as part of their antioxi dant enzyme system that helps the cell to counteract oxidative stress (36). A peroxidase mediated mechanism can explain the non-specific cytotoxicity of the tyrosinase inhibi tory agents we have tested. Putative depigmenting agents should subsequently lead to removal of unwanted pig ment when applied topically to skin. Thus, the effectiveness of deoxyarbutin as a treatment to reverse hyperpigmentation of human skin grafted to immunocompromised mice was assessed and compared with hydroquinone and 4-tertiary butylphenol (TBP). Xenografting of human skin onto immunocompromised mice is an excellent model for studying various aspects of skin physiology including hyperpigmentation of grafted skin on burn patients (3 7). In addition, this model is more physiologically relevant in that it allows the evaluation of melanogenic inhibitors directly on human skin. Hyperpigmen tation is common sequelae of most injuries to the skin (3 7). Farooqui et al. (3 7) showed that hyperpigmentation of human cutaneous xenografts placed on athymic nude mice was apparent as early as 4-6 weeks post-grafting. In addition, tyrosinase, a key enzyme in melanin biosynthesis, was upregulated in the grafted skin from 2 to 12 weeks post-grafting (3 7). Thus, topical application of a tyrosinase inhibitory agent could be an approach to reverse hyperpigmentation of the grafted skin and thus test its effect as a general skin-lightening modality. Topical application of dA reversed the skin hyper pigmentation of human skin grafted onto immunocompromised mice. Initial statistical analysis showed that treatments are statistically different within a week (p 0.0001). By statistically comparing the mean values for 11L * to the control group values, only one treatment group (HQ) at one time point (eight weeks) was statistically significant from the control group. Although skin lightening for dA, TBP, and even HQ at the other time points was not statistically significant from that of the control group, lightening
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)