312 JOURNAL OF COSMETIC SCIENCE triolein to be solubilized in such films (34). In other words, if the surfactant films were less ordered (e.g., if the hydrocarbon chains were of nonuniform length and/or branched), solubilization of triolein would be facilitated. Addition of short-chain alcohols is known to mitigate formation of undesirable liquid crystal phases (5,7,18,19,23). Joubran and coworkers (18) have also demonstrated that the formation of triglyceride microemulsion can be achieved by incorporating sucrose and a short-chain alcohol. The synergistic interactions among the alcohol and the sucrose molecules result in the destabilization of the liquid crystalline mesophases and thus facilitate the triglyceride microemulsion formation. They have also studied a micro emulsion of soybean oil, polyxyethylene (40) sorbitanhexaoleate, and water-ethanol and found that the microemulsion formation strongly depends on temperature and that the systems require large amounts of surfactants and alcohol (19). Von Corswant and co workers (5, 7) observed the presence of lamellar liquid crystal or L c phases in long-chain triglyceride systems. They explained that the L c is destabilized by incorporating water when a short-chain alcohol is present. Adding the short-chain alcohols can increase the flexibility of the surfactant film, since the L c phase is destabilized in favor of a micro emulsion phase due to the increase of the curvature (short-chain alcohols decrease the polarity of the aqueous phase). In addition, they found that adding co-oil into micro emulsion systems helps reduce the surfactant concentration and the amount of alcohol required· to form a microemulsion (7). Huang and Lips (11) also found that microemul sification of triglycerides requires high temperatures and surfactant concentrations, :whereas Minana-Parez and coworkers (35,36) have previously reported the use of alkyl sulfates with oxyethylene and oxypropylene groups, or so-called "extended surfactants," for forming efficient microemulsions of the bicontinuous type (Type III microemulsion) with polar oil, including triglycerides however, high salt concentrations (up to 7% wt NaCl) were required. The solubilization at optimum formulation with conventional surfactants (commercial ized non-extended surfactants) has been found to reach values as high as 30 ml/ml or ml/g surfactant with short-chain alkanes, and as high as 10 ml/g with hexadecane, while it can be less than 4 ml/g with mono-chain polar oils and much less with triglyceride oils (3 5 ). Graciaa and coworkers (3 7 ,38) first introduced the lipophilic linker concept to help enhance surfactant-oil interaction and thus improve the solubilization capacity of hydrocarbon and polar oils. Lipophilic linkers, such as a long-chain alcohol, tend to segregate near the oil side of the oil-water interface, close to the tails of the surfactants (39), as depicted in Figure 2. In this schematic, the surfactant, sodium dihexyl sulfo succinate, adsorbs at the oil-water interface. The lipophilic linker, dodecanol, is shown to adsorb at the palisade layer of the interface (oil side of the surfactant layer), promoting the local order and increasing the interaction between the surfactant tail and the oil phase. In contrast to the surfactant and the lipophilic linker, sodium mono- and di methyl naphthalene sulfonate (SMDNS) is a hydrophilic linker that segregates near the water side of the oil-water interface this hydrophilic linker molecule is believed to increase the total interfacial area and the overall interaction between the surfactant layer and the aqueous phase (40). For certain oils, adding a lipophilic linker alone to the microemulsion gives limited solubilization enhancement. Acosta and coworkers (39-41) found that hydrophilic linkers can help improve solubilization ability because they allow more room for lipophilic linkers to segregate and further enhance the solubilization ability.
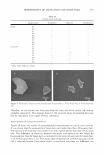
MICROEMULSIONS OF TRIGLYCERIDE-BASED OILS Water side of the interface Surfactant SDHS Sl.JNa SO 3 N a a � -;:,,-s '.:.��:�;:,, --;:::-0:,�;:,,;.0;._;;., � � � f Hydrophilic linker SMDNS Oil side of the interface Lipophilic linker Dodecanol SDHS = sodium dihexylsulfosuccinate Combined linker SMDNS = sodium mono- and dimcthylnaphalene sulfonate 313 Figure 2. Schematic of the linker concept, showing the surfactant (e.g., sodium dihexyl sulfosuccinate or SDHS), lipophilic linker (e.g., dodecanol), and hydrophilic linker (e.g., sodium mono- and di methylnaphthalene sulfonate or SMDNS). There are only limited choices of surfactants/ingredients that can be used in cosmetic formulations and that form microemulsions with triglycerides successfully. The main benefit of alcohol-free formulations stems from a consumer perception that alcohols are drying and potentially irritating, and so alcohol-free formulations would be useful for sensitive-skin consumers. This paper describes our successful use of linker concepts to formulate alcohol-free microemulsions with artificial sebum (human oil) using biocom patible and cosmetically friendly ingredients at ambient temperature and low salt con centrations. We have investigated the opportunity to tune the curvature of surfactant film by adding co-oil to the triglyceride in order to help triglyceride microemulsification as well as to minimize the surfactant required to form single-phase microemulsions without adding alcohol. EXPERIMENT AL PROCEDURES MATERIALS The following materials were obtained from Aldrich (Milwaukee, WI) at the concen trations shown and were used without further purification: sorbitan monooleate (Span 80, 99%), squalene (98%), squalane (99%), isopropyl myristate (IPM, 98%), ethyl laurate (99% ), and sodium chloride (99% ). Sodium dioctyl sulfosuccinate (AOT, -100%) was purchased from Fisher Scientific (Fair Lawn, NJ). Hexylpolyglucocide AG 6206™, donated by Akzo Nobel (Chicago, IL), was received as a 75 wt% aqueous
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)