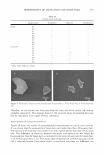
310 JOURNAL OF COSMETIC SCIENCE INTRODUCTION Microemulsions are transparent and thermodynamically stable mixtures of oil and water stabilized by surfactants. Microemulsions contain extremely high oil/water interfacial areas, offering ultra-low interfacial tension (less than 0.1 mN/m). Practical applications of microemulsion systems include enhanced oil recovery (EOR), drug delivery, nano particle synthesis, food, and cosmetics ( 1-3 ). The transparency of microemulsions makes them especially attractive for cosmetic formulations as they give the perception of a "clean" system. The ultralow interfacial tension between oil and water facilitates the penetration of the product into nanoscale pores of human skin, making microemulsions a candidate for deep-cleansing products. A Type I microemulsion (0/W microemulsion) is conceptualized as swollen micelles surrounded by water where surfactant micelles coexist with excess oil. A Type II mi croemulsion (W /0 microemulsion) is conceptualized as swollen reverse micelles sur rounded by oil where the reverse micelles coexist with excess water. A Type III micro emulsion consists of an oil, water, and a "middle" bicontinuous microemulsion phase coexisting in a three-phase equilibrium. A Type IV microemulsion is defined as a single-phase microemulsion system where both oil and water are completely solubilized in the surfactant microemulsion phase. Microemulsion transition can be achieved in several ways, depending on the type of surfactants. For example, for ionic surfactant systems, a Type I-III-II transition can be obtained by increasing the electrolyte concen tration, whereas increasing the temperature can achieve the same transition for nonionic surfactant systems. The optimum condition is defined as the condition at which an equal volume of oil and water is solubilized in the bicontinuous phase (Type III). The elec trolyte concentration required at the optimum condition is called "optimum salinity" or S*. The solubilization parameter (SP), which is defined by the amount of oil solubilized in the middle phase per unit mass of surfactant, at this optimum condition is known as the optimum solubilization parameter (SP*) maximizing this parameter for triglyceride oils is a goal of this work. Two important parameters that describe the ability and effectiveness of a surfactant to form microemulsions are the size and curvature of the microemulsion and the flexibility of the surfactant film it forms (4). An elastic and flexible surfactant film favors the formation of a microemulsion, whereas a lamellar phase is formed with a more rigid or stiff film. The flexibility of the film also depends on the molecular structure of the surfactant. Cosolvents such as short chain alcohols can improve the film's flexibility (5-8). While microemulsion phase behavior can be described in various ways, the "fish diagram" is one of the most common. A fish diagram is typically plotted between the surfactant concentration and a scan or tuning parameter (e.g., salt or hydrophobicity of the system), as shown in Figure 1. R0 is radius of the oil droplet and Rw is the radius of the water droplet. The curvature of the oil and water droplets is then equal to l/R0 and 1/Rw, respectively. The scan parameter directly affects the curvature of the surfac tant membrane, which is a very important factor for a surfactant to form microemul sions, as mentioned above. Electrolyte addition to ionic surfactant systems increases the hydrophobicity of the surfactant system and decreases the surfactant film curvature (see Figure 1). Therefore, when the surfactant system has relatively low hydrophobicity or is at low salinity, a Type I microemulsion (0/W microemulsion) occurs. At high hydrophobicity, where the cur-
MICROEMULSIONS OF TRIGLYCERIDE-BASED OILS Type IV: Single-phase microemulsion Type I: 1continuo Type II: Reverse micelles 311 ': ': '\ '/ \\ \\\/ :·::,:,:·::::,:::··::·:::::: � Salinity Curvature ( H)= 1 /R + 0 Reducing Curvature Figure 1. Fish diagram showing phase behavior and changes in curvature with surfactant concentration and formulation hydrophobicity as adjusted by a scanning variable (salinity). vature decreases, a Type II microemulsion (W/0 microemulsion) exists. When the hydrophobicity is intermediate between these two conditions, and at lower surfactant concentration, a three-phase microemulsion or Type III microemulsion occurs, with a net zero curvature. When the surfactant concentration increases within the Type III region, a Type IV microemulsion can be obtained. The minimum surfactant concentra tion for complete solubilization of the water and the oil is where the three-phase and one-phase regions (Type IV) meet, which appears at relatively high surfactant concen trations. Fish diagrams with similar behavior have been reported elsewhere (1,5,7,9-12). For example, Jakobs et al. (9) obtained the well-known fish diagram for water-n-decane nonionic surfactant systems by plotting surfactant concentration versus the scanning variable of temperature. Von Corswant and co-workers (5 ,7) plotted fish diagrams between alcohol concentration as a function of surfactant concentrations for triglyceride microemulsification. Formation of microemulsion systems with short-chain oils or alkanes has been exten sively studied (9,13-15) a large range of surfactants and additives can be used to control their microstructural properties. However, microemulsification of triglycerides, and in particular long-chain triglycerides, is very challenging (5-8,10,16-33). These triglyc erides have minimum solubility in microemulsion systems and thus tend to form liquid crystal mesophases. This is due to the fact that the hydrocarbon chain portions of the surfactant films present at triolein-water interfaces have difficulty penetrating the large triolein molecules. Therefore, higher temperatures are required to induce sufficient disorder of the triglyceride-surfactant film, which generally contains long hydrocarbon chai�s, straight and of uniform length. This disorder permits significant amounts of
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)