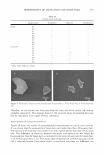
322 JOURNAL OF COSMETIC SCIENCE 60 --------------------------------, 50 -- -- tl -- -- -- 40 / Cl � C \ Jg 30 .... (,) -- ' � ' ::I !!!. 20 I I ♦ 10 0 0 0.2 0.4 0.6 0.8 1 Sebum fraction in oil Figure 7. Fish diagram with elthyl laurate (EL) at different salinities (the solid line and dashed line represent 0.5% and 1.5% NaCl, respectively) as a function of surfactant concentration and sebum fraction in oil (a value of O is 100% co-oil and 1 is 100% sebum oil). Surfactant/linkers studied here are AOT (4%), hexylglucoside (5.06%), and sorbitan monooleate (5.13%). The concentration ratio is kept constant as the total surfactant/linker concentration is varied (25°C). Refer to Figure 3 for the phase behavior at 0.5% NaCl and Figure 4 for the phase behavior at 1.5% NaCl. High-salinity systems create Winsor Type I-III-II and I-IV-II trans1t1ons at low and high surfactant concentrations, respectively. Neither nonmicroemulsion nor sponge phases are present. In addition, the fish seems to be more vertical than the fish at low salt concentrations, indicating less partitioning of surfactants or linkers into the excess phases. EFFECT OF TYPE OF CO-OIL ON THE FISH DIAGRAM The effect of the type of co-oil on phase behavior can be considered in terms of the change in the concentration, C, and the fraction of sebum in oil, F, as shown in Table IV. Changes in the C and F values at 0.5% NaCl are not significant when the hydro philicity of the co-oil increases from squalene to ethyl laurate. However, a decrease in the F value at 1.5% NaCl is observed: the F values are 0.2, 0.175, and 0.02 for squalane, IPM, and EL, respectively. Figure 8 shows the effect of the type of co-oil on the phase behavior at 1.5% NaCl the comparison is made only between squalane and ethyl laurate due to the relatively clear difference between the two types of co-oils. The result for ethyl laurate at 1.5% NaCl is of interest: the fish diagram appears at a very low sebum fraction in the mixed oil, or the F value is very close to zero, as shown in Figure 8. In other words, if F = 0, a Type II microemulsion is observed over the entire range of the sebum fraction in the mixed oil, as compared to the results with squalane (where F = 0.2). As mentioned earlier, this surfactant/linker system is relatively hydrophilic, creating the positive cur-
MICROEMULSIONS OF TRIGLYCERIDE-BASED OILS 323 50 � 40 0 30 ::I � 20 10 0 0 0.2 0.4 0.6 0.8 1 Sebum fraction in oil Figure 8. Fish diagram with squalane (solid line) and ethyl laurate (EL) (dashed line) at 1.5% NaCl as a function of surfactant concentration and sebum fraction in oil (a value of O is 100% co-oil and 1 is 100% sebum oil). Surfactant/linkers studied here are AOT (4%), hexylglucoside (5.06%), and sorbitan monooleate (5 .13% ). The concentration ratio is kept constant as the total surfactant/linker concentration is varied (25 ° C). Refer to Figure 4 for the phase behavior at 1.5% NaCl. vature with pure squalene at 0.5% NaCl, as shown in Figure 3. The addition of 1.5% NaCl makes the system become more hydrophobic and helps decrease the curvature of the surfactant membrane. Using co-oil that has a lower EACN, such as ethyl laurate, further decreases the curvature. As seen in Figure 8, a Type III microemulsion is observed in the absence of the sebum oil (the sebum fraction is zero). As the hydro philicity of the oil increases by increasing the sebum fraction in the mixed oil, the interaction between surfactant and oil is enhanced. This leads to a negative curvature, and the Type II microemulsion is formed. For the system with squalane, which is a more hydrophobic oil compared to ethyl laurate, a higher fraction of sebum oil is required to obtain the same curvature obtained from the system with ethyl laurate. CONCLUSIONS We have demonstrated that biocompatible and cosmetically friendly surfactants/linkers can be used to form single-phase triglyceride microemulsions (Winsor Type IV) at room temperature and low salt concentrations. The fish diagram observed in this study follows the classical patterns of phase behavior established for nonionic oligoethylene glycol based surfactants. We have also found that using co-oil helps the solubilization of triglycerides at lower surfactant concentrations and reduces the amount of surfactant necessary to form a single-phase microemulsion, which is desirable in cosmetic appli-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)