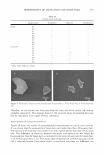
320 JOURNAL OF COSMETIC SCIENCE sitions are also observed at 1.5% NaCl for the systems with IPM and EL as co-oil, as well as at 3.0% NaCl for the system with squalane (data not shown). For the system at 0.5% NaCl, Winsor I-IV-I is observed at high surfactant concentration, as shown in Figure 3. As mentioned earlier, the phase behavior at 0.5% NaCl for squalene and squalane are similar, and so we can qualitatively compare the phase behavior for the system at low salt concentrations (studied with squalane) to the phase behavior at high salt concentrations (studied with squalane). For the system at 0.5% NaCl, as illustrated by the slanted fish in Figure 3, as the surfactant concentration increases, the surfactant system in the middle phase becomes more hydrophilic since more hydrophilic oil is required to form the middle phase (the fish head slants towards less hydrophilic oil). This suggests that the surfactant system requires higher salinities at higher surfactant concentrations to push the system towards the Type III or II microemulsions. Since the salt concentration is constant at 0.5% NaCl, this amount of salt is not sufficient to push the system towards Type II, and thus a Type I system is observed instead. Figure 5 shows the effect of salinity on phase behavior for the systems with squalane as co-oil. Both the body and the tail of the fish are observed for all three salinities: 0.5%, 1.5%, and 3.0% NaCl. As mentioned above, the phase behavior with squalane is similar to that for squalene (Figure 3 ), as seen in the following ways: (i) nonmicroemulsion is observed at a low surfactant concentration and a high sebum fraction in oil (ii) a Winsor � 0 � � � ::I rn 60 50 40 30 20 10 0 0 0.2 0.4 0.6 0.8 1 Sebum fraction in oil Figure 5. Fish diagram with squalane at different salinities (the thin solid line, dashed line, and thick solid line represent 0.5%, 1.5%, and 3% NaCl, respectively) as a function of surfactant concentration and sebum fraction in oil (a value of 0 is 100% co-oil and 1 is 100% sebum oil). Surfactant/linkers studied here are AOT (4%), hexylglucoside (5.06%), and sorbitan monooleate (5.13%). The concentration ratio is kept constant as the total surfactant/linker concentration is varied (25°C). Refer to Figure 3 for the phase behavior at 0.5% NaCl and Figure 4 for the phase behavior at 1.5%.
MICROEMULSIONS OF TRIGLYCERIDE-BASED OILS 321 Type I-III-II trans1t10n occurs at low surfactant concentrations, and a Winsor Type I-IV-I transition occurs at high surfactant concentrations when the sebum fraction in the oil increases and (iii) a Winsor Type I-IV-II transition appears at intermediate surfactant concentrations. As the salt concentration increases, the surfactant concentration at which Type IV forms, as denoted by "C," increases, whereas the fraction of sebum in the mixed oil ("F") at this point decreases. A similar trend is also seen for the system with IPM and EL, as shown in Figures 6 and 7. The values of C and F for the systems with different co-oil and different salinities are shown in Table IV. It is commonly known that adding salt increases the hydrophobicity of the surfactant system for a given co-oil, an increase in salinity shifts the phase behavior from Type I-III-II. When the surfactant system becomes more hydrophobic, the surfactant system can microemulsify a more hydrophobic oil. This is consistent with the observation in this paper that when salt concentration increases from 0.5% to 3.0%, the Type III micro emulsion forms at a lower sebum fraction in the mixed oil, which is more hydrophobic than the oil mixture with a higher sebum fraction. The concentration, C, increases with increasing salinity. This can be explained by the fact that adding salt in general decreases the solubilization because the salt molecules adsorb at the interface and displace the surfactant molecules, reducing the overall number of interactions per unit area (1). This behavior is also similar to that of the system in which low-molecular-weight alcohol is used. 60 50 ' ' '#. 40 � C: ca 30 rs/ 20 10 0 0 ::( � ' ' ""' ,;a' / l. \ J 0.2 _,;-'J , -- --er- , -- - 0.4 0.6 0.8 1 Sebum fraction in oil Figure 6. Fish diagram with isopropyl myristate (1PM) at different salinities (the solid line and dashed line represent 0.5% and 1.5% NaCl, respectively) as a function of surfactant concentration and sebum fraction in oil (a value of O is 100% co-oil and 1 is 100% sebum oil). Surfactant/linkers studied here are AOT (4%), hexylglucoside (5.06%), and sorbitan monooleate (5.13%). The concentration ratio is kept constant as the total surfactant/linker concentration is varied (25°C). Refer to Figure 3 for the phase behavior at 0.5% NaCl and Figure 4 for the phase behavior at 1.5% NaCl.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)