365 SYNTHESIS OF BENZYL ACETATE At the end of the reaction, ester and water are formed as products. With the formation of the ester, the catalyst becomes free. All steps are basic and irreversible, and the stage of final product formation is the rate-determination stage (Eq. 5) (50,51). C H CH B CH COOA H OH C H CH CH COO H O IL 6 5 2 3 6 5 2 3 2 + + + → + + + - (5) PROCEDURE OF ESTERIFICATION REACTION Esterification experiments were conducted in a 100 mL three-necked round-bottom flask glass reactor. Esterification of AA and BA catalyzed by IL experiments were conducted in a 100 mL three-necked round-bottom flask glass reactor. The reaction was carried out by effec- tive mixing by adding simultaneously certain amounts of AA (20 mmol), BA (20 mmol), and IL (10 mmol) (for AA:BA:IL molar ratio of 1:1:05) to the preheated reactor. The reactor was placed in a temperature-controlled oil bath with a mixer heater with a contact ther- mometer (IKA C-MAG HS7, IKA, Staufen, Germany.), and the reaction temperature was controlled. A reflux condenser was connected to the reactor to prevent evaporation losses during the reaction. The reaction mixture was effectively stirred with the magnetic stirrer. At the end of the reaction, a certain amount of sample was titrated with phenolphthalein indicator with 0.1 N NaOH, and acid conversion (%) was calculated by determining the amount of acid remaining in the phase without reacting (52–54). Trials were repeated at least twice for each case. The experiment was performed until reproducible results were obtained. The acid value (AV) was determined by Eq. 6. = × × L N f S T Acid Value mol (6) where N is the normality of NaOH, f is the factor of 0.1 N NaOH, S is the volume of the titrant (mL), and T is the volume of the sample (mL). The conversion (X) was calculated by comparing the AV of the initial acid (AV 0 ) to the AV of acid at time t (AV t ), using Eq. 7. (%) = - ×100 X AV AV AV t 0 0 (7) [EMIM] [HSO 4 ] was used as a catalyst in the esterification reaction biphasic layers are formed at the end of the reaction after cooling the solution to room temperature. The upper layer contains the desired ester product, while the bottom layer contains the IL, unreacted reactants, and water. The product (ester) can be easily removed from the reac- tion medium by decanting without any additional cost. Since the unreacted acid may be present in both phases, the acid analysis was carried out in both phases. [EMIM] [BF 4 ] was used as a catalyst in the reaction, the IL was precipitated as a solid, and the liquid phase consists of ester and unreacted reactants (55). The organic phase was analyzed to calculate the acid, which remained unreacted. [DEIM] [NTf 2 ], [EMIM] [NTf 2 ], and [OMIM] [BF 4 ] were used as the catalyst in the reaction, and the reaction medium was not biphasic because the ester was dissolved in the IL after cooling the solution to room temperature (56). In this case, the acid analysis was carried out in the reaction mixture.
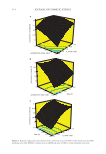
366 JOURNAL OF COSMETIC SCIENCE EXPERIMENTAL DESIGN AND BOX–BEHNKEN MODELING The response surface methodology is used to determine the mathematical relationship between the dependent variable (response) and the independent variables and to opti- mize the response variable affected by the various process parameters. This method also describes the effect of single or multiple combinations of independent variables on the process response (57). The Box–Behnken experimental design of response surface methodology was applied to estimate the conversion of AA (%) and reaction parameters. For this purpose, the Design-Expert® Software Version 7 Trial (Stat-Ease, Inc., USA) was used in which the experimental studies will be performed for the esterification reaction of AA with BA. In the study, the effect of three process parameters on acid conversion (%) was investigated to determine the catalyst activity of ILs. Box–Behnken experimental design was made with three levels and three factors. A total of 17 experiments were designed, including five center points. Three parameters affecting the esterification reaction, acid/alcohol mole ratio (x 1 ), IL mole ratio (x 2 ), and time (x 3 ), were determined as the process parameters in the system, and these parameters were cho- sen as independent variables. Variable levels and coded values were shown in Table I. The conversion of AA (%) was selected as the dependent variable (58). The response surface function of the parameters that affect the process is expressed as follows (59) (Eq. 8): ε ) (x = ….,x + Y f x , , k 1 2 (8) where Y is the dependent variable or response, x i is the independent variable, f is the function of response, and ε is the experimental error. The second-order polynomial equation (Eq. 9) represents the response surface method as shown in the following (60): ∑ ∑β ∑∑β β β ε = + + + + = =1 =1 =2 Y x x x x i k i i i k ii i i k-1 j k ij i j 0 1 2 (9) where β 0 is the constant regression coefficient, β i , β ii , β ij are the interaction coefficients, and k is the factor number. The statistical analysis of the experimental data was carried out with the Design-Expert program. Analysis of variance (ANOVA) was used to explain the effect of variables on the mathematical model obtained in the selected study range. Table I Variable Levels and Coded Values in the Experimental Design Range and level Independent variable Symbol –1 0 1 Acid/alcohol molar ratio x 1 1.0 1.5 2 IL molar ratio x 2 0.25 0.50 0.75 Reaction time (h) x 3 4 6 8
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)