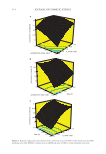
367 SYNTHESIS OF BENZYL ACETATE RESULTS AND DISCUSSION Time, temperature, catalyst ratio, and acid/alcohol mole ratio parameters were investi- gated in detail for the esterification experiments. Besides, the experimental design of the studies was conducted to determine the effects of the interactions of the variables in the system used separately and on each other. The effect of the presence and type of ILs used as catalysts in esterification reactions on acid conversion was investigated. For this purpose, firstly, esterification of AA and BA without catalyst was carried out according to the experimental procedure described. The obtained results of the conversion of AA (%) were presented in Figure 1. The rate of catalyst-free esterification reaction is prolonged under atmospheric conditions. The reaction rate depends on the rate of catalysis of the carboxylic acid itself. For a faster reaction, a catalyst is added to the reaction medium to serve as a proton donor to the car- boxylic acid (61). For this purpose, ILs consisting of [EMIM]+, [OMIM]+, and [DEIM]+ cationic groups and [HSO 4 ]−, [BF 4 ]−, and [NTf 2 ]− anionic groups were used as catalysts. EFFECT OF EXPERIMENTAL VARIABLES For industrial esterification reactions, the kinetic study of the conversion is of great importance for process design and economy. In order to investigate the esterification reaction, kinetics of AA with BA catalyzed by [EMIM] [HSO 4 ], experiments were done with AA:BA:[EMIM] [HSO 4 ] molar ratio of 1:1:0.5 at 110°C. While only 39.59% of AA was converted to benzyl acetate at the end of 1 h, this conversion increased to 85.3% at the end of 8 h, and the system came to equilibrium at the end of this period. The effect of time on acid conversion (%) was shown in Figure 2. It can be seen that the conversion of AA will not change after this time. For this reason, 4, 6, and 8 h, which are the critical stages of the conversion, were chosen as the reaction time in the optimization experiments performed in the later stages of the study. Generally, in esterification reactions, high temperature increases the reaction rate and acid conversion (62). Therefore, three different temperatures were selected to find the 2 1.5 1 0 20 40 60 80 100 4 6 8 Reaction time (h) Figure 1. Conversion of acid (%) for acetic acid and benzyl alcohol esterification without catalyst at 110°C. Conversion of acid (%)
368 JOURNAL OF COSMETIC SCIENCE highest acid conversion for the esterification reaction system. The boiling temperatures of the reactants and the product (benzyl acetate) were taken into consideration when mak- ing the selection. In order to investigate the effect of temperature on the esterification reaction of AA with BA catalyzed by [EMIM] [HSO 4 ], experiments were performed for 8 h with AA:BA:[EMIM] [HSO 4 ] molar ratio of 1:1:0.75 at 90–100°C and 110°C. The results were shown in Figure 3. It has been determined that 110°C is the reaction tem- perature at which the conversion of acid close to 90% is achieved. The effect of AA to BA molar ratio on the esterification reaction was investigated exper- imentally. Similar to previous studies in the literature, AA:BA molar ratio of 1, 1.5, 2 was selected. The experiments at 4, 6, and, 8 h in which [EMIM] [HSO 4 ] mole ratio was 0.75 were carried out at 110°C. The results were given in Figure 4. It was seen that as the number of acid increases, the conversion in the reaction decreases. It shows that when the 0 20 40 60 80 100 0 1 2 3 4 5 6 7 8 Reaction time (h) Figure 2. Effect of time on the conversion of acid (%) using [EMIM] [HSO 4 ] as catalyst. (AA: BA: [EMIM] [HSO 4 ] molar ratio 1:1:0.5, T=110°C) 0 10 20 30 40 50 60 70 80 90 100 90 100 110 Temperature (°C) Figure 3. Effect of temperature on the conversion of acetic acid (AA) (%) using [EMIM] [HSO 4 ] as catalyst. (Reaction conditions: AA: BA: [EMIM] [HSO 4 ] molar ratio 1:1:0.75, t = 8 h) Conversion of acid (% ) Conversion of acid (%)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)