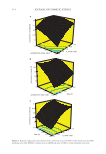
369 SYNTHESIS OF BENZYL ACETATE initial acid concentration is excess in the reaction medium, the reversible esterification reaction is more effective in the reverse direction. Therefore, the conversion is low due to a large amount of product formed. Conventional acidic catalysts used in esterification reactions cause corrosion of process equipment and pose a threat to environment. Experiments were carried out to get an idea about the amount of ILs used as catalysts to eliminate these disadvantages. In this study in which high conversion was aimed using an optimum catalyst, the relationship between the change of IL amount and conversion AA:BA:[EMIM] [HSO 4 ] molar ratio was investigated by experiments between 1:1:0.15 and 1:1:0.75. Figure 5 showed that AA conversion increases with an increasing amount of IL in the reaction. For this reason, in optimization experiments, IL molar ratios were studied as 0.25, 0.50, and 0.75. 4 6 8 0 20 40 60 80 100 1 1.5 2 AA : BA molar ratio Figure 4. Effect of acid/alcohol molar ratio on the conversion of acid (%) using [EMIM] [HSO 4 ] as catalyst at T = 110°C. 0 10 20 30 40 50 60 70 80 90 100 0.00 0.15 0.25 0.50 0.75 IL molar ratio Figure 5. Effect of amount of catalyst on the conversion of acid (%) using [EMIM] [HSO 4 ] as catalyst. (Reaction Conditions: AA:BA = 1:1 T = 110°C, t = 6 h) Conversion of acid (%) Conversion of acid (% )
370 JOURNAL OF COSMETIC SCIENCE BOX–BEHNKEN EXPERIMENTAL DESIGN At this stage of the work, the studying range was determined by considering the prelimi- nary experiment and critical limit values of the process variables (time, acid/alcohol molar ratio, and IL molar ratio). These values were entered into the “Design Expert” program, and the experimental set was designed (Table II). Box–Behnken design, which is one of the most widely used methods for RSM in the lit- erature (63–66), was chosen as the experimental design method to be studied. The main objective of this method is to evaluate the many variables and to minimize the number of experiments required. In addition, process variables are examined and used to model the system’s response to these variables. Experiments were carried out according to the test procedure mentioned earlier, provided that the experimental conditions were the same for each catalyst. The conversion of acid (%) obtained with AA:BA:[EMIM] [HSO 4 ] molar ratio of 1:1:0.75 at 110° C for 6 h was presented in Figure 6. It was seen that 91% of acid was converted to benzyl acetate if [EMIM] [HSO 4 ] was used, 82% of acid was converted if [DEIM] [NTf 2 ] was used, 77% of acid was converted if [EMIM] [NTf 2 ] was used, 75% of acid was converted to benzyl acetate if [OMIM] [BF 4 ] was used, and 72% of acid was converted to benzyl acetate when [EMIM] [BF 4 ] was used. The comparative list of catalytic activity for the ILs was as follows: Table II The Experimental Design and Response Values for [EMIM] [HSO 4 ] Experiment number x 1 x 2 x 3 Conversion of acid (%) Y 1 1.0 0.25 6 77.96 2 1.0 0.50 4 85.59 3 1.0 0.50 8 85.30 4 1.0 0.75 6 91.00 5 1.5 0.25 4 63.50 6 1.5 0.25 8 71.44 7 1.5 0.50 6 80.40 8 1.5 0.50 6 79.96 9 1.5 0.50 6 79.13 10 1.5 0.50 6 79.27 11 1.5 0.50 6 79.44 12 1.5 0.75 4 83.96 13 1.5 0.75 8 87.54 14 2.0 0.25 6 42.82 15 2.0 0.50 4 68.58 16 2.0 0.50 8 76.94 17 2.0 0.75 6 77.92
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)