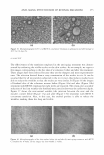
306 JOURNAL OF COSMETIC SCIENCE of the grafts was obvious both visually and from the gradual positive increase in '1L * compared to the vehicle-treated control. The increase in '1L * started for dA at the two-week treatment point and increased gradually over the eight-week treatment pe riod. In addition, TBP-treated grafts developed clear vitiligo-like patches. This discrep ancy may result from the high SE and low number of animals in each group. For instance, in order to be able to detect a significant difference for the dA-treated group eight weeks after treatment, using the two-sided two-sample test, we needed at least five animals in each group. The effect of dA on reversing hyperpigmentation demonstrated in the xenograft model system was recapitulated in a clinical trial. Hyperpigmentation induced by UV exposure was effectively reduced by dA. The lightening effect of dA on pathologic hyperpigmen tary disorders like melasma, solar lentigo, and post-inflammatory hyperpigmentation is yet to be determined. An interesting observation is that mice treated with HQ developed brown coloration of the albinistic hair adjacent to the grafts. Presumably this is a staining artifact from the by-products of hydroquinone oxidation. This brown pigmentation matches the reported nail plate discoloration that can develop during use of topical hydroquinone-containing cream (38--40). This color change of the nail was considered to be due to the oxidation products of hydroquinone. Hydroquinone readily oxidized to quinone and subsequently to hydroxyquinone, which is unstable and polymerizes to a brown compound. CONCLUSIONS In this report we have provided evidence that currently used skin-depigmenting agents have drawbacks in either their efficacy or their safety. Deoxyarbutin is demonstrated to be less cytotoxic than the standard skin-lightening product in the market (i.e., HQ). The cytotoxicity dA exhibited is not associated with dramatic degenerative changes in the morphology of keratinocytes as compared with HQ. Our overall goal is to provide the market with an effective skin depigmenting agent due to the fact that available depig menting agents in the market are far from satisfactory. We have clearly demonstrated that dA has the potential to be a safe and effective depigmenting agent. It also offers the potential to be an effective alternative to hydroquinone, the skin-depigmenting standard in the market with known safety drawbacks. REFERENCES (1) R. E. Boissy, M. Visscher, and M.A. deLong. DeoxyArbutin, A novel tyrosinase inhibitor with effective skin lightening potency, Exp. Dermatol., 14, 601-608 (2005). (2) J.P. Ortonne and]. J. Nordlund, Mechanisms that cause abnormal skin color, in The Pigmentary System: Physiology and Pathophysiology, J. J. Nordlund, R. E. Boissy, V. J. Hearing, R. A. King, and J. P., Ortonne, Eds. (Oxford University Press, New York, 1998), pp. 489-502. (3) A. Perez-Bernal, M.A. Mufioz-Perez, and F. Camacho, Management of facial hyperpigmentation, Am. ]. Clin. Dermatol., l, 261-268 (2000). (4) S. Briganti, E. Camera, and M. Picardo, Chemical and instrumental approaches to treat hyperpig mentation (review), Pigment Cell Res., 16, 101-110 (2003). (5) S. Passi and M. Nazzaro-Porro, Molecular basis of substrate and inhibitory specificity of tyrosinase: Phenolic compounds, Br.]. Dermatol., 104, 659-665 (1981).
EFFICACY AND SAFETY OF DEOXYARBUTIN 307 (6) K. B. Penney, C. J. Smith, and J.C. Allen, Depigmencing action of hydroquinone depends on dis ruption of fundamental cell processes,]. Invest. Dermatol., 82, 308-310 (1984). (7) A. Palumbo, M. D'ischia, G. Misuraca, and G. Prata, Skin depigmentation by hydroquinone: A chemical and biochemical insight, Pigment Cell Res., 2 (Suppl.), 299-303 (1992). (8) E. Serra-Baldrich, M. J. Trib6, and J. G. Camarasa, Allergic contact dermatitis from kojic acid, Contact Dermatitis, 39, 86 (1998). (9) M. Nakagawa, K. Kawai, and K. Kawai, Contact allergy to kojic acid in skin care products, Contact Dermatitis, 32, 9-13 (1995). (10) L. Petit and G. E. Pierard, Skin-lightening products revisited, Int.]. Cosmet. Sci., 25, 169-181 (2003). (11) Y. Hirn, S. Hatae, T. Inoue, and Y. Ohyama, Inhibitory effects of kojic acid on melanin formation: In vitro and in vivo studies in black goldfish,]. Jap. Cosmet. Sci. Soc., 6, 193-194 (1982). (12) Y. Ohyama and Y. Mishima, Melanogenesis-inhibitory effect of kojic acid and its action mechanism, Fragrance, 18, 53-58 (1990). (13) J. T. Lim, Treatment of melasma using kojic acid in a gel containing hydroquinone and glycolic acid, Dermatol. Surg., 25, 282-284 (1999). (14) A. Garcia and J.E. Fulton, The combination of glycolic acid and hydroquinone or kojic acid for the treatment of melasma and related conditions, Dermatol. Surg., 22, 443--447 (1996). (15) E. V. Curto, C. Kwong, H. Hermersdorfer, et al., Inhibitors of mammalian melanocyte tyrosinase: In vitro comparisons of alkyl esters of gentistic acid with other putative inhibitors, Biochem. Pharmacol., 57, 663-672 (1999). (16) T. Sugai, Clinical effects of arbutin in patients with chloasma (in Japanese), Hifu (Skin Res.), 34, 522-529 (1992). (17) A. K. Chakraborty, Y. Funasaka, M. Komoto, and M. Ichihashi, Effect of arbutin on melanogenic proteins in human melanocytes, Pigment Cell Res., 11, 206-212 (1998). (18) K. Maeda and M. Fukuda, Arbutin: Mechanism of its depigmenting action in human melanocyte culture,]. Pharmacol. Exp. Therapeutics, 276, 765-769 (1996). (19) M. Nakajima, I. Shinoda, Y. Fukuwatari, and H. Hayasawa, Arbutin increases the pigmentation of cultured human melanocytes through mechanisms other than the induction of tyrosinase activity, Pigment Cell Res., 11, 12-17 (1998). (20) H. Matsuda, S. Nakamura, H. Shiomoto, T. Tanak, and M. Kubo, Pharmacological studies on leaf of Arctostaphylos uva-ursi (1.) Spreng. IV. Effect of SO% methanolic extract from Arctostaphylos uva-ursi (1.) Spreng (bearberry leaf) on melanin synthesis. (inJapanese), Yakugaku-Zasshi.J 112, 276-282 (1992). (21) R. E. Boissy and P. Manga, On the etiology of contact/occupational vitiligo, Pigment Cell Res., 17, 208-214 (2004). (22) H. Zhao and R. E. Boissy, Distinguishing between the catalytic potential and apparent expression of tyrosinase activities, Am.]. Med. Sci., 308, 322-330 (1994). (23) S. Pomerantz, Tyrosine hydroxylation catalyzed by mammalian tyrosinase: An improved method of assay, Biochem. Biophys. Res. Commun., 16, 188-194 (1964.) (24) S. E. Papadakis, S. Abdul-Malek, R. E. Kamdem, and K. L. Yam, A versatile and inexpensive tech nique for measuring color of foods, Food Technology, 54, 48-51 (2000). (25) S. Passi, M. Picardo, and M. Nazzaro-Porro, Comparative cytotoxicity of phenols in vitro, Biochemj., 245, 537-542 (1987). (26) C. J. Smith, K. B. O'Hare, and J.C. Allen, Selective cytotoxicity of hydroquinone for melanocyte derived cells is mediated by tyrosinase activity but independent of melanin content, Pigment Cell Res., 1, 386-389 (1988). (27) N. Watanabe and H.J. Forman, Autoxidation of extracellular hydroquinones is a causative event for the cytotoxicity of menadione and DMNQ in AS49-S cells, Arch. Biochem. Biophys., 411, 145-157 (2003). (28) M. P. Tabibian, Skin lightening/depigmenting agents, eMedicine ]., 2, 1-7 (2001). (29) K. Jimbow, H. Obata, M.A. Pathak, and T. B. Fitzpatrick, Mechanism of depigmentation by hyd roquinone,J. Invest. Dermatol., 62, 436-449 (1974). (30) G. A. Gellin and H. I. Maibach, Chemically induced depigmentation, Models Dermatol., 2, 282-286 (1985). (31) K-1. Nihei and I. Kubo, Identification of oxidation product of arbutin in mushroom tyrosinase assay system, Bioorgan. Medicin. Chem. Lett., 13, 2409-2412 (2003). (32) B. Kasraee, Peroxidase-mediated mechanisms are involved in the melanocytotoxic and melanogenesis inhibiting effects of chemical agents (editorial), Dermatology., 205, 329-339 (2002).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)