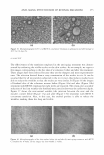
308 JOURNAL OF COSMETIC SCIENCE (33) H.B. Dunford and A. J. Adeniran, Hammet rho sigma correlation for reactions of horseradish per oxidases compound II with phenols, Arch. Biochem. Biophys., 251, 536-542 (1986). (34) D. C. Thompson, Y. N. Cha, and M.A. Trush, The peroxidases-dependent activation of butylated hydroxyanisole and butylated hydroxyroluene (BHT) to reactive intermediates: Formation of BHT quinone methide via a chemical-chemical interaction,]. Biol. Chem., 264, 3957-3965 (1989). (35) V. V. Subrahmanyam, P. Kolachana, and M. T. Smith, Metabolism of hydroquinone by human my eloperoxidase: Mechanisms of stimulation by other phenolic compounds, Arch. Biochem. Biophys., 286, 76-84 (1991). (36) J.J. Yohn, D. A. Norris, D. G. Yrastorza, I.J. Buno,J. A. Leff, S.S. Hake, and]. E. Repine, Disparate antioxidant enzyme activities in cultured human cutaneous fibroblasts, keratinocytes and melanocytes, ]. Invest. Dermatol., 97, 405--409 (1991). (37) J. Z. Farooqui, B. W. Auclair, E. Robb, E. Sarkisian, C. Cooper, J. W. Alexander, G. Warden, R. E. Boissy, and J. J. Nordlund, Histological, biochemical, and ultrastructural studies on hyperpigmented human skin xenografts, Pigment Cell Res., 6, 226-233 (1993). (38) R. J. Mann and R. R. M. Harman, Nail staining due to hydroquinone skin-lightening creams, Br.]. Dermatol., 108, 363-365 (1983). (39) I. H. Coulson, "Fade out" photochromonychia, Clin. Exper. Dermatol., 18, 87-88 (1993). (40) R. L. Garcia, J. W. White, and W. F. Willis, Hydroquinone nail pigmentation, Arch. Dermatol, 114, 1402-1403 (1978).
]. Cosmet. Sci., 55, 309-325 Quly / August 2006) Microemulsions of triglyceride-based oils: The effect of co-oil and salinity on phase diagrams NAPAPORN KOMESV ARAKUL, MONICA D. SANDERS, ERIKA SZEKERES, EDGAR]. ACOSTA, JAMES F. FALLER, TONY MENTLIK, LOUIS B. FISHER, GREGG NICOLL, DAVID A SABATINI, and JOHN F. SCAMEHORN, University of Oklahoma, School of Chemical, Biological, and Material Engineering and the Institute for Applied Surfactant Research, Sarkeys Energy Center, 100 E. Boyd, Norman, OK 73019 (N.K., M.D.S., E.S., J.F.S.), University of Toronto, Chemical Engineering and Applied Chemistry, 200 College Street, Toronto, Ontario, Canada M5S3E5 (BJ.A.), Mary Kay Inc., 1330 Regal Row, Dallas, TX 75247 U.F.F., T.M., L.B.F., G.N.), and University of Oklahoma, Civil Engineering and Environmental Science Department and the Institute for Applied Surfactant Research, 202 W. Boyd, Norman, OK 73019 (D.A.S.). Accepted for Publication March 2, 2006. Synopsis Microemulsification of triglyceride-based oil is challenging due to the formation of undesirable phases such as macroemulsions, liquid crystals, or sponge phases. This research evaluates the formation of artificial sebum microemulsions using linker molecules, with the addition of co-oil to help enhance sebum solubi lization. The microemulsion consists of a lipophilic linker (sorbitan monooleate), a hydrophilic linker (hexylglucocide), a main surfactant (sodium dioctyl sulfosuccinate), a co-oil, and artificial sebum. The effect of adding co-oil to the phase behavior and the microstructure of the resulting microemulsion is described. The effect of several types of co-oil is also studied the co-oils evaluated here are sgualene, sgualane, isopropyl myristate, and ethyl laurate. The effect of salinity on the microemulsion phase behavior is also presented. Fish diagrams are obtained by plotting total surfactant/linker concentration as a function of sebum fraction in the oil mixture (co-oil + sebum). Different microemulsion types (Winsor Types I, II, III, and IV) are formed, depending on the total surfactant/linker concentration and the fraction of co-oil in the oil mixture. Winsor Type IV (single-phase) microemulsions are observed at high surfactant/linker concentrations. These single-phase, isotropic, and low-viscous fluids are particularly useful for cleansing and delivery of functional ingredients in skin care products. Salt addition shifts the fish diagram towards more hydrophobic oil systems and higher surfactant/linker concentrations. The current address of Napaporn Komesvarakul is Unilever Home and Personal Care-North America, 40 Merritt Blvd, Trumbull, CT 06611. The current address of Erika Szekeres is Clorox Service Company, 7200 Johnson Drive, Pleasanton, CA 94588. Address all correspondence to David A. Sabatini. 309
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)