318 JOURNAL OF COSMETIC SCIENCE can be achieved in two ways: increasing the hydrophobicity of the surfactant system (aqueous phase) or increasing the hydrophilicity of the oil. Increasing the hydrophobicity of the surfactant system helps to move the surfactant system to the aqueous phase-oil phase interface, whereas increasing the hydrophilicity of the oil phase helps match the hydrophobicity of the surfactant system with the oil phase. The hydrophobicity/ hydrophilicity matching leads to an increase in penetration of the surfactant film into the oil phase, a decrease in the curvature from positive values (Type I) to negative values (Type II), and an increase in the flexibility of the film. As seen in these results, the Type I-III-II transition is obtained when the fraction of sebum in the oil increases (oil hydrophilicity increases). It is evident that the addition of sebum oil to the system induces a change in the microstructure of the microemulsion from an O/W type (Type I) to an W/O type (Type II). This suggests that the interaction between the surfactant film and the sebum oil is increased as the hydrophilicity of the oil mixture increases. From Figure 3, it is worth mentioning that less than 30 wt% surfactant is necessary to microemulsify triglyceride-based oil at room temperature. This is much less than in previous reports that required up to 50% surfactants and co-surfactants for triglyceride microemulsification (6,34). The required temperature in the microemulsification of sebum oil is also much lower than the temperatures that were reported in studies with triolein (ranging from 25° to 60°C in the presence and absence of alcohols, respectively (5 ,43 ). This is attributed to the presence of fatty acids in the sebum, which facilitate the oil solubilization (34, 11). Fish diagrams for the systems with squalane, isopropyl myristate, and ethyl laurate as co-oil at 0.5% NaCl are similar to the results shown in Figure 3 therefore, detailed description of these systems is not provided. Von Corswant et al. (5,7) found that adding isopropyl myristate to microemulsions based on triglycerides decreased the spontaneous curvature of the surfactant film and increased flexibility of the surfactant monolayer. The change in spontaneous curvature was manifested by a gradual change in the microstruc ture of the microemulsion, as revealed by NMR self-diffusion data. Interestingly, a Type I-III-II transition for a long-chain triglyceride was observed when the amount of IPM increased, whereas an opposite trend is observed here that is, a Type I-III-II transition occurs when the sebum fraction in oil increases or when the fraction of IPM in the oil mixture decreases. This might be due to the fact that the surfactant that Von Corswant et al. used, which is soybean phosphatidylcholine (SbPC), is relatively hydrophobic thus when the oil mixture is relatively hydrophobic, the degree of surfactant-oil interaction increases. This is known to decrease the curvature and increase the flexibility of the film, inducing a Type I-III-II transition. This is also supported by the findings of Von Corswant et al. the water/1-propanol/SbPC/IPM microemulsion system forms a W/O or Type II microemulsion (that is, the spontaneous curvature of the SbPC film is negative). In the system reported here, the increased hydrophilicity of the sebum and co-oil mixture enhances the surfactant-oil interaction, leading to a Type I-III-II transition as well when the amount of IPM present is reduced. In other words, the spontaneous curvature for the surfactant film investigated here is positive when co-oil is present alone. Surfactant partitioning at the excess water/middle phase and the middle phase/excess oil interfaces. When surfactant concentration (y-axis) is plotted as a function of a tuning parameter such as salinities or hydrophobicity (x-axis), a fish diagram typically appears to be vertical in both fish body and fish tail. This suggests an insignificant partitioning of lipophilic and hydrophilic compounds from a bicontinuous middle phase into an excess
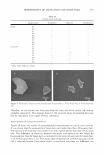
MICROEMULSIONS OF TRIGLYCERIDE-BASED OILS 319 oil phase and an excess water phase, respectively. In some cases, the head of the fish can slant towards lower salt concentration when the surfactant concentration decreases. In this case, as the surfactant concentration increases, the middle-phase microemulsion requires higher salinities i.e., the surfactant system in the middle phase becomes more hydrophilic, suggesting that the lipophilic compound present in the middle-phase partitions into the excess oil. In contrast, if the head of the fish slants towards higher salt concentrations, the middle phase becomes more hydrophobic, which can be attributed to the partitioning of the hydrophilic compound into the excess water. This well known phenomenon is described in Bourrel and Schecter (1). As shown in Figure 3, the fish leans towards high-hydrophobicity oil when the sebum fraction in oil is close to zero or when the fraction of co-oil is equal to one. In other words, when surfactant concentration increases, the middle-phase microemulsion requires more hydrophilic oil. This suggests that the middle phase becomes more hydrophilic, implying the partitioning of the lipophilic compound, which is sorbitol monooleate, into the excess oil phase. EFFECT OF SALINITY ON THE FISH DIAGRAM FOR SQUALANE, ISOPROPYL MYRISTATE, AND ETHYL LAURA TE A fish diagram with squalane at 1.5% NaCl is shown in Figure 4. The fish diagram at this salt concentration varies as compared to the fish diagram at 0.5% salt concentration (Figure 3). At 1.5% NaCl, Winsor I-III-II and I-IV-II transitions are observed at low and high surfactant concentrations, respectively. These Winsor I-III-II and I-IV-II tran- 60 IV 50 � 40 C: 30 II 20 : 10 a 0 0.2 0.4 0.6 0.8 1 Sebum fraction in oil Figure 4. Fish diagram with squalane at 1.5% NaCl as a function of surfactant concentration and sebum fraction in oil (a value of O is 100% co-oil and 1 is 100% sebum oil). Surfactant/linkers studied here are AOT ( 4% ), hexylglucoside (5 .06% ), and sorbitan monooleate (5 .13 % ). The concentration ratio is kept constant as the total surfactant/linker concentration is varied. (25°C).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)