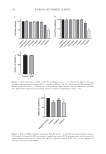
119 A New Alternative for Acne Treatment EE OF THE TEXTILE MATERIALS TREATED WITH BPO MICROSPONGES To determine the EE and DL, fabric samples (4 cm x 4 cm) were dissolved in 50 mL acetone at 200 rpm for 48 hours. To determine the amount of active substance loaded in the fabric samples, the solution was filtered through a 0.22 µm syringe filter, and filtrate was analyzed by a validated UPLC method. Each batch was evaluated five times. The EE was determined by using Eq. 4: EE% Experimental drug content Theoretical drug content = ×100 (4) IN VITRO DRUG RELEASE OF THE TEXTILE MATERIALS TREATED WITH BPO MICROSPONGES To determine in vitro drug release profiles of the textile materials treated with BPO microsponges, fabric samples (4 cm x 4 cm) were placed in glass vessels containing 100 mL of phosphate buffer solution+acetone (4:1) solution at 37 °C and 70 rpm. At appropriate time intervals, samples were withdrawn, and the amount of BPO released was determined by a validated UPLC method. The experiment was carried out three times. RESULTS AND DISCUSSION Microsponges were successfully obtained by the quasi emulsion solvent diffusion method. A validated UPLC method was used to measure the concentration of BPO from micro- sponges. The UPLC method was developed and validated following the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use guide- lines’ recommendations (Q2) for validation of analytical procedures (24). The method showed that BPO peaked at 3.67 minutes, and the chromatogram is shown in Figure 2. BPO was successfully separated from other compounds. The calibration curve of BPO was linear in the concentration range of 0.5–10 µg/mL (r2 = 0.9996). The limit of detection and the limit of quantification of BPO were found to be 0.242 and 0.403 µg/mL, respectively (24). The EE and DL of BPO were as high as 60.879% and 38.929%, respectively. Considering the high EE, it can be concluded that this method is a simple and suitable technique for producing BPO-loaded microsponges. The morphology of the microsponges were investigated by SEM analysis. It was seen that the microsponges were uniform and had a spherical shape. The presence of surface pores could be clearly observed. The SEM images of the optimized microsponge formulation are shown in Figure 3. The particle size and distribution analysis were performed, and the particle size (Dv50) of the selected formulation was found to be 111 µm with a 2.567 span value. Dv50, defined as the median particle size by volume, is the particle diameter of 50% of the sample volume. A particle size distribution graph is shown in Figure 4, and the values are given in Table II. The FTIR patterns were evaluated in order to understand whether there were any interactions between the drug and the polymer. The FTIR patterns of BPO, EC, and BPO microsponges are shown in Figure 5.
120 JOURNAL OF COSMETIC SCIENCE Figure 3. SEM micrographs of BPO microsponges at different magnifications. Figure 4. Particle size distribution of microsponges. Table II Particle Size and Distribution Values of the Microsponge Dv10 Dv50 Dv90 Span 38.1 µm 111 µm 323 µm 2.567 Figure 5. Fourier transform infrared spectra of pure BPO, EC, and BPO-loaded microsponges.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)