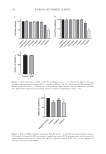
178 J. Cosmet. Sci., 73, 178–189 (May/June 2022) Address all correspondence to John S. Bajor, john.bajor@unilever.com The Safety and Efficacy of Petrolatum CAROL A. BOSKO, JEAN ADAMUS AND JOHN S. BAJOR Unilever Research, 40 Merritt Blvd, Trumbull, Connecticut, USA (J. B., C.B., Ret., J.E., Ret.) Accepted for publication February 23, 2022. HISTORY Petroleum jelly (PJ) was invented by Robert Chesebrough, a chemist, who was first inspired to investigate the properties of petroleum “wax” during a visit to the oil fields of Titusville, Pennsylvania in 1859. Chesebrough noticed that oil workers were using the waxy substance that accumulated at the top of the oil drills to help heal their cuts and burns. He brought the substance back to his lab in Brooklyn, New York, developed a procedure to purify the material, and undertook an effort to identify its utility. In 1865, Chesebrough applied for his first patent describing a triple refining process that created the PJ, or petrolatum, that we know today (1). So ardent was his belief in his “wonder jelly” that Chesebrough traveled around New York selling the semisolid from a horse-drawn cart as he purportedly burned himself. He then slathered the jelly onto his fresh wounds in front of crowds that had gathered to see his demonstrations. It was not until 1872 that Chesebrough dubbed his product Vaseline® (Unilever, Trumbull, Connecticut), from the German name for water (wasser) and the Greek word for oil (oilon) (2). The main advantage of Vaseline PJ (VPJ) versus other vegetable-based oils that were in use at the time was its stability and resistance to oxidation and hydrolysis. VPJ won numerous awards, including the grand medal at the Philadelphia Exposition of 1876 and the silver medal at the Paris Exposition of 1878. By the late 1880s, the product was so successful that production could not keep up with demand, and shortages occurred in 1885 and again in 1891 (3). At that time, Vaseline and Epsom salts shared the number one spot as the most prescribed products in a survey of pharmacists. Chesebrough survived to the age of 96 and attributed his longevity to eating a spoonful of VPJ every day (4). COMPOSITION PJ is an odorless, colorless, translucent, and semisolid mixture of hydrocarbons with a melting point ∼37°C. It is a purified derivative of petroleum, containing a liquid hydrocarbon fraction, paraffin wax, and a microcrystalline wax (5,6). Paraffin wax contains
179 Efficacy of Petrolatum mostly straight-chain alkanes of 26 to 30 carbon atoms, while the microcrystalline wax contains isoalkanes and napthene-containing alkanes of 41 to 50 carbons. Petrolatum is a viscoelastic material (both fluid and elastic properties) owing to the presence of partly crystalline lamellar sheets that trap the liquid fraction (7). Paraffin oil can be comprised of hundreds of different species, and its composition and physical properties are largely dependent on processing (6). In fact, the safety and efficacy of petrolatum is highly dependent on the refinement process (8). VPJ is the original branded version of petrolatum, and it is refined to a very high standard that removes unwanted impurities. SAFETY VPJ has been safely used for almost 150 years and is widely used in both cosmetic and pharmaceutical preparations (7). Petrolatum is listed as an active ingredient in the Skin Protectant Monograph for over-the-counter drugs (9) and must meet specification set by the FDA and the European Pharmacopeia (10 and references therein). Petrolatum products that meet purity tests as published in the Code of Federal Regulations are permissible in food products (FDA regulation #21CFD172.880). Thus, petrolatum is safe if ingested only when it is refined to the appropriate standards. PJ is compatible with all skin types, is fragrance-free, is nonirritating (11), is hypoallergenic, and is noncomedogenic (12,13). Despite its occlusive properties and lipidic sensories, PJ can be used to moisturize acne-prone skin without exacerbating this condition (12). Allergic reactions to petrolatum are rare because the saturated hydrocarbons are not easily oxidized or metabolized, and it is frequently used as the vehicle in irritancy, allergenicity, photoallergy, and phototoxicity testing (14). There is no evidence of cumulative irritation potential. VPJ causes no adverse reactions in skin, not only due to its high standard of processing, but also because it remains in the stratum corneum. Numerous studies have shown that the hydrocarbons contained in petrolatum remain almost entirely in the stratum corneum, with few reaching the epidermis or dermis, even when applied on acetone-treated skin (15,16). A review of in vitro and in vivo penetration studies of petroleum derivatives used in cosmetics showed no evidence of systemic exposure from topically application (10). Polycyclic aromatic hydrocarbon levels in petrolatum are very low, as mandated by the FDA regulations cited previously. Numerous studies have failed to demonstrate any toxicity or carcinogenicity of PJ (8). Petrolatum is among the most highly tested materials in the world and has a long history, which confirms the safety of this material both for topical use and by ingestion. UTILITY IN DERMATOLOGY PJ helps promote skin health and healing in persons with compromised skin, including dry, cracked, and chapped skin (hands, heels, lips, angular chilitis, diaper rash) atopic dermatitis (AD) eczema and certain medical conditions. It is endorsed as a post-procedure treatment for minor wound healing and is typically considered the first line of defense against ingress of certain microbes and from external insults. Once considered nonphysiologic, VPJ is not completely inert in fact, compared to other skin treatments, it provides several additional benefits for skin. These include skin barrier recovery acceleration (15,17), preventing infections postoperatively, and stimulating the innate immune response (17).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)