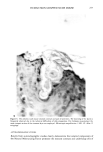
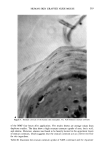
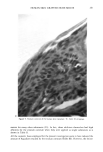
CLEAR GEL ANTIPERSPIRANTS 231 CONCLUSION Solid antiperspirant gelled sticks can be prepared using DBS as the gelling agent and anhydrous alcohols and glycols as the solvent if suitable stabilizing agents are em- ployed. Zinc acetate, methenamine (hexamethylene tetramine), and acetamide MEA have been identified as suitable stabilizing agents. REFERENCES (1) E. L. Roehl and H. B. Tan, to Naarden International N.V., Solid antiperspirant composition and process for its preparation, US 4154816 (1979). (2) E. L. Roehl, to Naarden International N.V., Solid antiperspirant composition and process for its preparation, US 4346079 (1982). (3) P. Thomas and M. Sibi, Organogels obtained from the benzoic acetal of sorbitol, Cornpt. Rend., 183, 282-284 (1926). (4) J. K. Wolfe, R. M. Hann, and C. S. Hudson, 1,2,3,4-dibenzylidene-D-sorbitol,J. Am. Chem. Soc., 64, 1493-1497 (1942). (5) P. J. Van Der Laan and L. P. Van Der Mijll Dekker, The constitution of monobenzylidene-D-sor- bitol, Rec. Tray. Chim., 62, 824-826 (1943). (6) L. Vargha, Constitution of monobenzal-D-sorbitol. Remarks on a communication of P. J. Van Der Laan and L. P. Van Der Mijll Dekker, Ber., 77B, 608 (1944). (7) S. J. Angyal and J. V. Lawler, 1,3:2,4-dibenzylidene-D-sorbitol, J. Am. Chem. Soc., 66, 837-838 (1944). (8) S. Yamamoto, Sorbitol derivatives. IV. Purity of benzalsorbitol, ibid., 51, 114-115 (1948). (9) H. Hagiwara, Chemical structure of dibenzylidenesorbitol, ibid., 72, 929-930 (1952). (10) J. E. Courtois and P. Le Dizet, Preparation of L-xylose after periodic acid oxidation of o-benzylidene- sorbitol, Bull. Soc. Chim. Biol., 37, 287-289 (1955). (11) C. Ukita and S. Suzuki, Structure of 2,3,4,5-di-o-benzylidene-D-glucose derivatives, Yakugaku Zasshi, 81, 225-226 (1961). (12) T. G. Bonner, E. J. Bourne, and D. Lewis, 2,4:3,5-di-o-benzylidene-D-glucitol, Carbohyd. Res., 2, 421-425 (1966). (13) J. Kovar, Z. Burianec, and J. Jary, Solvolysis of benzylidene derivatives. III. Dibenzylidene deriva- tives of alditols, Collect. Czech. Chem. Commun., 32, 4099-4107 (1967). (14) T. G. Bonner, E. J. Bourne, P. J. V. Cleare, and D. Lewis, Kinetic and thermodynamic control in the formation of monoacetals from aldehydes and D-glucitol, J. Chem. Soc. B, 827-830 (1968). (15) D. J. Brecknell, R. M. Carman, J. J. Kirby, and L. T. Nicholas, Tri-o-benzylidene-D-Glucitol, Aust. J. Chem., 29, 1859-1863, (1976). (16) J. March, Advanced Organic Chemistry.' Reactions, Mechanisms, and Structure (McGraw-Hill, New York, 1968), pp 661-663. (17) T. J. Schamper, M. M. Perl, and J. D. Warren, to American Cyanamid Co., Acid stable dibenzyl monosorbitol acetal gels, US 4518582 (1985).
j. Soc. Cosmet. Chem., 37, 233-247 (July/August 1986) Sensory segmentation of fragrance preferences HOWARD R. MOSKOWITZ, Moskowitz/_Jacobs, inc., Valhalla, New York Received November 8, 1985. Presented at the Annual Scientific Meeting of the Society of Cosmetic Chemists, New York, December 5-6, 1985. Synopsis This paper illustrates a method by which one can segment consumers into distinct segments of individuals showing different fragrance preferences. By testing various in-market fragrances and prototype fragrances "blind," and instructing panelists to profile these fragrances on a variety of acceptance and sensory charac- teristics, one can uncover the underlying segments. This study reveals three segments--a low impact segment, a medium impact segment (likers of sweet and floral notes), and a high impact segment (likers of spicy, heavy fragrances). The procedure also illustrates how the same sensory attribute generates quite different patterns of acceptance, depending upon the sensory segment. INTRODUCTION Of all the senses, our senses of smell and taste are most pervaded by individual differ- ences in liking (1). Smells that one person likes another person might dislike very much. Indeed, to many researchers the variation in acceptance of a fragrance from one person to another is sufficient cause to abandon the search for regularity in olfactory perception and hedonics (1-3). Much of what we know about smell comes from psychophysics, the study of sensory processes (4). Traditionally, psychophysicists have preferred to study well-defined model systems, such as pure chemicals (5,6). Even with these model systems, the vari- ability of hedonics emerges (7-9). However, there exists relatively little in the psycho- physics and sensory research which points to generalities in odor likes and dislikes. We know that people like the smell of roses and hate the odor of carbon bisulfide, but there are precious few generalities (10). The literature on odor hedonics and individual differences is equally barren when we look in the perfumery and cosmetics literature. For one, most researchers working for private companies cannot publish the results of their research (11). Second, there has been little in the way of a general approach to inter-individual differences in percep- tions. Perfumers recognize these differences and live with them. They develop fra- grances to fit target demographic or psychographic segments. Presumably these con- sumer segments should exhibit fragrance preferences. That assumption remains to be tested, however. THE CONCEPT OF SEGMENTATION Over the past twenty years marketers have increasingly recognized that the consumer world does not comprise a monolithic structure with identical preferences governing each consumer. Rather, there are segments in this group, with each segment presumed 233
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)