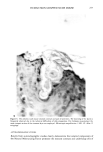
FALSE POSITIVE NITROSAMINE ANALYSES 271 NMDDA 100.0 - 43 50.0 74 211 61 198 - I , , ,, I II, 111 168 I .... • ,', '1 .... t .... [" "•'" '1 .... I .... I' "'1" "1" "•' "'1 ""l' •' I .... • .... I 60 80 1 O0 120 140 160 180 200 220 M/Z 40 Figure 2. Electron impact mass spectrum of a standard 88 ng/•l NMDDA solution 4 •l splitless injec- tion. only selected ions specific for a compound(s) of interest. Figure 3a shows a series of these single ion chromatograms called "mass chromatograms" from scan 1700-2100 for a 0.5 •1 splitless injection of the shampoo extract at masses 198 and 211, indicative of NMDDA, and mass 228, which is the molecular weight of myristic acid. Figure 3b shows a 1 •1 splitless injection of the same shampoo extract which was spiked with 3 •1 of an 88 ppm NMDDA standard. Note the appearance of a new peak in the single ion chromatograms for masses 198 and 211 (at scans 1861/1862) which were not present in the 198 and 211 single ion chromatograms of the neat shampoo extract (Figure 3a). Although the NMDDA standard does coelute with the myristic acid, the single ion monitoring capability makes it possible to observe the NMDDA in the spiked sample which does not appear in the neat shampoo extract. A new peak also appeared at scan 2026 (mass 198) due to an impurity in the NMDDA standard. As a further confirmation that the shampoo extract did not contain NMDDA and that the TEA signal was a "false positive," a UV-photolysis confirmatory experiment was performed on the shampoo extract. Nitroso compounds (e.g., N-nitrosamines) are not stable to UV radiation while nitro compounds are stable (8). Upon irradiation of the extract followed by reanalysis with the GC/TEA, disappearance of the TEA response indicates the presence of a nitroso compound while no change in the TEA signal indi- cates the presence of a nitro compound (8). UV irradiation of the shampoo extract did not alter the TEA signal. This observation supports the GC/MS results indicating no NMDDA was present in the shampoo extract.
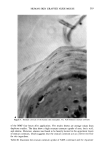
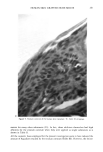
272 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Shampoo Extract a. 7.6 198 5.7 211 100.0 228 1700 15:35 1885 1884 1796 i I I I [ I I I • 1881 1929 I ! 1929 ! I 4 i I I I I I [llllllll'l • 1928 i ] I , , i I ' I [ , I i , , [ I I I"] , [ I [ I [ I I I , i [ i ß . , I 1881 1831 1743 ,, ,,1,8• 1..•928 52 ',, , , ,, ,, ,,q-,,,, ,2•_08, ,, ,2Or__.. 1750 1800 1850 1900 1950 2000 2050 2100 SCAN 16:02 16:30 16:57 17:25 17:52 18:20 18:47 19:15 TIME b. 10.4 198 Shampoo Extract Spiked With NMDDA 100.0 228 1861 I 8 . . ß ' I ' ' ' ' I ' " ' ' [ ' ' ' ' I ' ' ' ' I '[ ' '[ ' I ' ' • [ [ I ] ] 33.8 1862 211 I 1831_• 1.7•.9,.7 1929 ,2q7,2 [ ß ß ' [ .... "• ' ' ' ! ' ' ' ' ! ' •' ' I ' ' ' ' I ' • ' ' I •' ' I 1881 , _ _ _• 1927 ' ' ' ' .... ' ' ' ß ' ' ' ' [ ' "• ' [ ' ' ' ' I ' ' ' ' ] [ [ ' ' 1881 • 19.,.•9_29 2003 2063 I ''' ' ' I .... ! ' ' ' ' ! ' ' ' ' [ " ' ' ' I ' ' ' ' •-' ' ' ' I I I ! I 1700 1750 1800 1850 1900 1950 2000 2050 2100 SCAN 15:35 16:02 16:30 16:57 17:25 17:52 18:20 18:47 19:15 TIME Figure 3. (a) Mass chromatograms for a 0.5 txl splitless injection of a shampoo extract at masses 198 and 211, indicative of NMDDA, and mass 228, the molecular weight of myristic acid. Also shown is the reconstructed ion chromatogram. (b) Mass chromatograms and reconstructed ion chromatogram (as de- scribed above) for a 1 txl splitless injection of the same shampoo extract spiked with 3 txl of a standard 88 ng/txl NMDDA solution. Note the appearance of a new peak at scans 1861/1862.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)