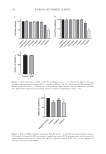
135 PHYSALIS ANGULATE CREAM FOR SOLAR MELANOSIS pain and complications such as persistent hyperemia, postinflammatory hyperchromia, and hypochromia (8). Controlling the postIPL inflammatory process can optimize the aesthetic results since the inflammatory modulators, such as prostaglandins E2 and interleukin-1, favor the formation of erythema and increased production of metalloproteinase enzymes (MMPs), which induce the degradation of cellular matrix proteins. MMPs are important enzymes in all phases of healing. However, when not properly modulated, these enzymes can impair the production of collagen and elastin, compromise the process of tissue regeneration, and cause treatment- opposite effects (23). In order to treat skin inflammation, especially after IPL application, hydrocortisone was the first topical glucocorticoid to show efficacy and has been widely used to provide comfort to patients. However, hydrocortisone use incurs unwanted side effects (24). As a result, anti- inflammatory substances of natural origin with minimal side effects have been studied frequently (22–25). Medicinal plants can produce several types of compounds through alterations in their natural habitat, secondary metabolism, and production of active principles (26–29). P. angulate has been indicated as an alternative treatment of the inflammatory process after IPL application this is due to its anti-inflammatory properties and lack of any apparent side effects (19–26). In addition, it has antinociceptive (pain relief) effects (19) this is observed in this study as none of the G2-PA and only 16.6% of the G1-PA participants reported pain. In a study, 0.5 mg of topical physalin E (extracted from aerial parts of P. angulate) was compared to a topical corticosteroid (0.5 mg dexamethasone) in the treatment of chronic and acute dermatitis in the ears of rats. Physalin E reduced edema in 39% of animals, reduced tumor necrosis factor-α, and inhibited myeloperoxidase. Although not comparable to dexamethasone, physalin E showed a potential anti-inflammatory effect (15). Similarly, in the present study, only four participants (13.3%) of G1-PA had edema compared to three participants (10%) who presented with edema in the hydrocortisone group. In another study, physalin B, C, and G, purified from P. angulate extract, reduced nitric oxide production through macrophages that were stimulated by lipopolysaccharides and interferon-γ. Physalin B produced lower levels of TNF-α, interleukin-6, and interleukin-12 in.stimulated macrophages. This inhibition caused by physalin B was not affected by the antiglucocorticoid, RU-486 ([4-dimethylamino]phenyl-17beta-hydroxy- 17-[1-propynyl]estra-4,9-dien-3-one), suggesting that physalin B acts using a different mechanism than corticosteroids. Physalin B, F, and G have also shown in vitro efficacy in preventing transplant rejection (30). The alcoholic extract of P. angulate demonstrated the ability to decrease the production of inflammatory cytokines, interleukin-1 alpha and interleukin-6, histamine, and prostaglandin E2 in an in vitro study. These molecules lead to the emergence of symptoms that characterize acute phase systemic responses including temperature elevation (31). Moreover, the alcoholic extract of P. angulate can increase the production of interleukin-10, which is an important anti-inflammatory cytokine (20) that may be related to the absence of temperature elevation in the G2-PA group. Moreover, one of the clinical signs of the acute inflammatory response is characterized by erythema, which was absent in G2-PA.
136 JOURNAL OF COSMETIC SCIENCE Presence of polyunsaturated fatty acids (omega-3, omega-6, and omega-9) was revealed previously in the same study. Polyunsaturated fatty acids have an important role in the restoration of skin barrier healing because they modulate inflammatory responses (32). Dermatological procedures such as IPL, laser, and peeling can cause damage to the skin surface, which compromises the skin barrier function as these methods remove epidermal cells (33–35). There is evidence that the skin barrier is metabolically active and participates in inflammatory responses through the activation of cytokines, fibroblasts, melanocytes, and new vascular formation (36). Omega-9 fatty acid can significantly inhibit the production of nitric oxide, improve the level of interleukin-10, and decrease the expression of cyclooxygenase-2 (COX-2) in the skin lesion (37–39). It is possible that the improvement of inflammation through P. angulate does not happen only by using physalins and phytosteroids. The present study showed similar results to those of previous studies conducted using P. angulate. G1-PA results were less effective than G1-H. In contrast, G2-PA showed better results than G2-V such as improvements in all objective and subjective parameters including pain, erythema, and temperature. The temperature in 28 participants (93.3%) was higher in the hand that was treated with the moisturizing extract of P. angulate, and in 13 participants (43.3%), it was 1°C. CONCLUSION This study shows that the cream containing 0.5% P. angulate L. extract may be useful when a mild anti-inflammatory agent is required for treating skin-related inflammations. Although the response was lower compared to hydrocortisone, these clinical results highlighted the anti-inflammatory properties of the cream and its potential as a dermocosmetic. This study also paves the way for further research involving preclinical studies with different concentrations to prove the benefits and quantify the anti-inflammatory action of P. angulata in comparison to that of corticosteroids. REFERENCES (1) S. Cignachi, V. Campos, L. Maluf, L. Grohs, M. Wancizinski and M. Costa, Comparative study of the effectiveness of 2940-nm, 1340-nm laser and intense pulsed light use on global rejuvenation of hands. J Am Acad Dermatol, 72, 1–267 (2015). (2) A. Kawada, H. Shiraishi, M. Asai, H. Kameyama, Y. Sangen, Y. Aragane and T. Tezuka, Clinical improvement of solar lentigines and ephelides with an intense pulsed light source. J Dermatol Surg, 28 (6), 504–508 (2002). (3) H. Sasaya, A. Kawada, T Wada, A. Hirao, and N. Oiso, Clinical effectiveness of intense pulsed light therapy for solar lentigines of the hands. J Dermatol Surg, 24 (6), 584–586 (2011). (4) R. C. R. Patriota, C. J. Rodrigues, and L. C. Cucé, Luz intensa pulsada no fotoenvelhecimento: Avaliação clínica, histopatológica e imuno-histoquímica. An Bras Dermatol, 86 (6), 1129–1133 (2011). (5) M. G. Cunha, F. D. Paravic and C. A. Machado, Alterações histológicas dos tipos de colágeno após diferentes modalidades de tratamento para remodelamento dérmico: Uma revisão bibliográfica. Surg Cosmet Dermatolo, 7 (4), 285–292 (2015). (6) S. A. P. Sampaio and E. A. Rivitti, Dermatologia. 3rd Ed. (Artes Medicas, São Paulo, 2008).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)