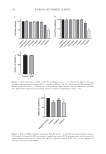
140 J. Cosmet. Sci., 73, 140–153 (May/June 2022) Address all correspondence to Thomas P. Dooley, tom@tomdooley.org Formulation Development of Sustained-Release Microencapsulated IR3535® with Long-Duration Mosquito Repellency EDUARD CASAS-TERRADELLAS, EMMA BEBBINGTON, TYCHO SPEAKER AND THOMAS P. DOOLEY LivFul Inc., Cheshire, United Kingdom (E.C.T., E.B., T.P.D.) Capsulent, Santa Cruz, California, USA (T.S.) Accepted for publication June 10, 2022. Synopsis Bites from pathogen-harboring insects (e.g., mosquitos and sandflies) and noninsect arthropod pests (e.g., ticks) can result in a variety of vector-borne pathogenic diseases. Numerous commercial repellent products containing DEET, IR3535® (Merck KGaA, Darmstadt, Germany), or other repellent active ingredients—but lacking sustained-release technology—are effective at repelling pests from human skin for several hours. To reduce or prevent the potential for arthropod vector-borne diseases, sustained release formulations of repellent active ingredients with long-duration benefits are needed to increase efficacy and user compliance. Here, the authors present the formulation development of a Lewis acid–Lewis base adduct encapsulated emulsion containing 20% w/w IR3535. Stability testing of the lotion was performed at ambient room temperature (20°–25°C) and 40°C. Repellency efficacy on human subjects was tested against Aedes aegypti mosquitos in laboratory arm-in-cage studies. Anti-infective properties were assessed against multiple bacterial species. A series of sustained-release lotions of microencapsulated IR3535 was prepared. During formulation development, chemical stability was problematic. However, chemical stability was achieved by buffering the lotion with sodium citrate–citric acid. The microencapsulated formulation manifested a long- duration efficacy of 12–13 hours against A aegypti mosquitos in laboratory arm-in-cage studies. The lotion also demonstrated anti-infective properties against multiple bacterial species. This sustained release formulation achieved a long-duration efficacy benefit superior to commercial repellent products, without the unpleasant odor or oily feeling that is customary for DEET-containing products. INTRODUCTION Insect-borne and non-insect arthropod pest-borne pathogenic diseases affect millions of people worldwide with acute and chronic conditions. Some of the prominent vector- mediated infectious diseases include malaria, dengue fever, Lyme disease (borreliosis), leishmaniasis, Chagas disease (trypanosomiasis), Zika fever, equine encephalitis, and yellow fever. In lieu of (or in addition to) eradication of, or physical separation from, insects and noninsect arthropod pests, prevention of vector-borne diseases or the symptoms thereof
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)