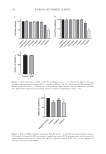
141 MICROENCAPSULATED INSECT REPELLENT may be achieved by application to the skin, hair, and/or clothing of an effective long- duration sustained-release repellent formulation. The repellency efficacy of a topical formulation is a function of: (a) the active ingredient concentration on the skin at a particular time (b) the rate of skin penetration and (c) the rate of aromatic evaporation from the skin, in addition to the arthropod species and environmental conditions. An ideal sustained release formulation will reduce the rate of skin penetration and evaporation rate of the active ingredient maintain a residual concentration of the active ingredient and prevent the active ingredient from evaporating without entirely sequestering it. Thus, a formulation chemist seeks a “slow” rate of evaporation rather than “no” rate, whereas nonsustained-release conventional formulations exhibit rapid and high volatility without long-duration efficacy. Sustained-release formulations can fail during development for various reasons, including the three parameters mentioned previously, as well as chemical instability of the active ingredient and/or excipients and physical instability (phase separation, flocculation, viscosity variations, etc.). Achieving an effective topical sustained-release formulation can be challenging, as one must optimize the composition in conjunction with the manufacturing processes (e.g., the sequence of addition of the ingredients, temperatures, and mixing intensity). Microencapsulation of the common synthetic active ingredient DEET within a topical repellent formulation has been demonstrated to decrease the rate of skin (“transdermal”) penetration (1,2). Microencapsulation can also prolong evaporation rates of DEET, thus providing a sustained release mode of action (2). These publications, in conjunction with a related US patent T.J. Speaker, Microencapsulation product and process, US Patent 8,039,015 (2011), have disclosed the microencapsulation of an emulsion using Lewis acid–Lewis base adduct interactions. In the present work, the authors desired to use a similar Lewis acid– Lewis base microencapsulation approach, albeit using another active ingredient, IR3535®. This ingredient has several properties that make it more appealing than DEET to consumers. IR3535 has been known for more than 2 decades to be an effective and safe mosquito repellent active ingredient. This active ingredient has been included in multiple commercial consumer products in various countries. The efficacy of IR3535-containing topical formulations (e.g., sprays and lotions) against mosquitos has been ranked relative to DEET-containing formulations (3–10). Comparisons of the two active ingredients have also been made using the insect vectors of Chagas disease (trypanosomiasis), (11,12), lone star ticks (Amblyomma americanum) (13), and two species of sandflies (Phlebotomus mascittii and Phlebotomus duboscqi), which are the vectors of leishmaniasis (14). A third species of sandflies, Phlebotomus papatasi, has also been tested for IR3535 (15). In the context of typical commercial formulations, in repellency efficacy comparisons of the same or similar concentrations of active ingredients, IR3535 is less effective or comparable to DEET. For example, the mean complete protection time (CPT) for human subjects in a field trial in the Florida Everglades for 25% IR3535 in ethanol was 3.0 hours versus 5.6 hours for 25% DEET (4). The CPT is the number of minutes (or hours) elapsed between topical application and the first landing and/or probing (biting) of the biting insect. The CPT may be defined as the time of the confirmed first bite (CFB), which represents the first subject within a group of subjects experiencing his/her second bite or confirmatory bite. Although these publications disclose the duration of repellency efficacy for compositions containing IR3535, they do not present comparisons of standard formulations (e.g., in
142 JOURNAL OF COSMETIC SCIENCE ethanol or an emulsion) versus sustained-release formulations that are intended to enhance long-duration repellency efficacy. The repellency of DEET can be greater than that of IR3535. However, the latter has several distinct advantages over DEET for use on human or nonhuman mammalian skin Most notably, IR3535 lacks DEET’s strong odor and produces a nonoily/nongreasy feel for preferred cosmetic appeal to consumers. Still, a repellent formulation containing IR3535 with high mosquito repellency (e.g., 12+ hours on human skin) has not been disclosed in prior studies or for commercial products. A major goal while developing a topical consumer product (e.g., insect repellent lotion) is the establishment of a stable composition with a “formal” and/or “imputed” shelf life of at least 12 months, preferably 18 months, and more preferably 24 or 36 months at ambient room temperature (RT) (20°–25°C). Inherent within this shelf life are multiple relevant parameters, such as: (a) overall chemical stability, an example of which is pH stability, preferably exhibiting no more than 1.0 pH unit variance (decrease or increase) during the imputed or formal shelf life and (b) active ingredient stability with a variance of no more than 5% to 6% loss of the active ingredient during the imputed or formal shelf life. In other words, if there is an initial nominal active ingredient concentration of 20% w/w, then the loss over time must not exceed 0.05 x 20% w/w or 0.06 x 20% w/w. Thus, a loss greater than 1.0% or 1.2% respectively of the nominal concentration of the product label is unacceptable (i.e., a concentration less than 19.0 or 18.8% w/w respectively). Whenever sufficient time does not permit the formal establishment of a shelf life, extrapolations of linear regression slopes obtained over shorter time periods at accelerated temperatures—such as 40°C and/or 50°C—can be used as surrogates to establish an “imputed” shelf life. When comparing RT of approximately 20°–25°C to 40°C, the shortened time interval’s result at elevated temperature is extrapolated or multiplied by a factor based upon empirical results. One may observe that 40°C accelerates the instability or other failure aspect(s) of the formulation (e.g., reduction in active ingredient, phase separation, flocculation, viscosity changes, etc.) over RT by a factor of, for instance, 3 times. Thus, in this example, if it does not fail at 6 months at 40°C, but fails shortly thereafter, then the imputed RT shelf life would be limited to 6 months times 3, or 18 months. International regulatory agencies state the allowed tolerances or variances for the content of the active ingredient to be maintained during the entire shelf life of insect repellents or pesticides. WHO guidelines declare an allowed tolerance of 6% for repellent products with a declared nominal content of active ingredient at 20°C ± 2 of 10% to 25% w/w or w/v (16). This 6% tolerance is also accepted by the European Chemicals Agency and most of the other regulatory authorities. However, the US Environmental Protection Agency (EPA) specifies an active ingredient tolerance of only 5%. Thus, the variance from the nominal label amount or concentration of the active ingredient should not exceed 5% or 6% over the designated shelf life of the repellent product. As an example, a product with a nominal concentration of 20% w/w of active ingredient should not have a concentration greater than 21% w/w or less than 19% w/w using the EPA tolerance standard, or a slightly higher tolerance in most jurisdictions outside of the United States. The authors describe the formulation development and properties of a repellent lotion consisting of 20% w/w IR3535 in a Lewis acid–Lewis base adduct for microencapsulation and carefully selected cosmetic ingredients.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)