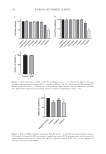
163 ANTIPIGMENTATION AND ANTIOXIDANT ACTIVITY CONCLUSION It is well-known that UV exposure can trigger oxidative stress, and the superfluous ROS accumulation promotes melanin synthesis and activates tyrosinase in melanocytes. In this study, SFE effectively alleviated UVB-induced oxidative stress by increasing GSH-Px and T-AOC activities in epidermal keratinocytes. It also exhibited potent inhibitory activity on melanin production caused by UVB exposure in a 3D skin model, which suggested that it might have a similar whitening effect on human skin. Above all, it is suggested that SFE will expand its applications in cosmetic manufacture. ACKNOWLEDGMENTS This work was supported by the Yunnan Science and technology project (2018ZF005). REFERENCES (1) H. Liu, X. Lu, Y. Hu, and X. Fan, Chemical constituents of Panax ginseng and Panax notoginseng explain why they differ in therapeutic efficacy, Pharmacol. Res., 161, 105263 (2020). (2) B.R. Yang, S.C. Yuen, G.Y. Fan, W.H. Cong, S.W. Leung, and S.M. Lee, Identification of certain Panax species to be potential substitutes for Panax notoginseng in hemostatic treatments, Pharmacol. Res., 134, 1–15 (2018). (3) L. Peiran, L. Ying, Z. Mingzhuo, Y. Ye, and C. Xiuming, The development of a Panax notoginseng medicinal liquor processing technology using the response surface method and a study of its antioxidant activity and its effects on mouse melanoma B16 cells, Food Func.., 8(11), 4251–4264 (2017). (4) H. Dumbuya, S.Y. Hafez, and E. Oancea, Cross talk between calcium and ROS regulate the UVA- induced melanin response in human melanocytes, FASEB J., 34(9), 11605–11623 (2020). (5) J.O. Lee, E. Kim, J.H. Kim, Y.H. Hong, H.G. Kim, D. Jeong, J. Kim, S.H. Kim, C. Park, D.B. Seo, Y.J. Son, S.Y. Han, and J.Y. Cho, Antimelanogenesis and skin-protective activities of Panax ginseng calyx ethanol extract, J. Ginseng. Res., 42(3), 389–399 (2018). (6) Y.C. Hseu, Y. Vudhya Gowrisankar, L.W. Wang, Y.Z. Zhang, X.Z. Chen, P.J. Huang, H.R. Yen, and H.L. Yang, The in vitro and in vivo depigmenting activity of pterostilbene through induction of autophagy in melanocytes and inhibition of UVA-irradiated α-MSH in keratinocytes via Nrf2-mediated antioxidant pathways, Redox Biol., 44, 102007 (2021). (7) S.H. Hashemi-Shahri, A. Golshan, S.A. Mohajeri, J. Baharara, E. Amini, F. Salek, A. Sahebkar, and Z. Tayarani-Najaran, ROS-scavenging and anti-tyrosinase properties of crocetin on B16F10 murine melanoma cells, Anticancer Agents Med. Chem., 18(7), 1064–1069 (2018). (8) J. Kim, Y.H. Kim, S. Bang, H. Yoo, I. Kim, S.E. Chang, and Y. Song, L-765,314 suppresses melanin synthesis by regulating tyrosinase activity, Molecules, 24(4), 773 (2019). (9) S.Y. Shim, Y.E. Lee, and M. Lee, Antioxidant compounds, kirenol and methyl ent-16α, 17-dihydroxy- kauran-19-oate bioactivity-guided isolated from Siegesbeckia glabrescens attenuates MITF-mediated melanogenesis via inhibition of intracellular ros production, Molecules, 26(7), 1940 (2021). (10) A.R. Im, S.H. Yeon, J.S. Lee, K.A. Um, Y.J. Ahn, and S. Chae, Protective effect of fermented Cyclopia intermedia against UVB-induced damage in HaCaT human keratinocytes, BMC Complement. Altern. Med., 16, 261 (2016). (11) A.Y. Lee, Skin pigmentation abnormalities and their possible relationship with skin aging, Int. J. Mol. Sci., 22(7), 3727 (2021). (12) Y. Zi, B. Zhang, B. Jiang, X. Yang, Z. Liang, W. Liu, C. He, and L. Liu, Antioxidant action and protective and reparative effects of lentinan on oxidative damage in HaCaT cells, J. Cosmet. Dermatol., 17(6), 1108–1114 (2018). (13) T. Fu, B. Chai, Y. Shi, Y. Dang, and X. Ye, Fargesin inhibits melanin synthesis in murine malignant and immortalized melanocytes by regulating PKA/CREB and P38/MAPK signaling pathways, J. Dermatol. Sci., 94(1), 213–219 (2019).
164 JOURNAL OF COSMETIC SCIENCE (14) I.M.N. Molagoda, W. Karunarathne, S.R. Park, Y.H. Choi, E.K. Park, C.Y. Jin, H. Yu, W.S. Jo, K.T. Lee, and G.Y. Kim, GSK-3β-Targeting fisetin promotes melanogenesis in B16F10 melanoma cells and zebrafish larvae through β-catenin activation, Int. J. Mol. Sci., 21(1), 312 (2020). (15) Y. Wang, T. Duan, M. Hong, Y. Zhou, H. Huang, X. Xiao, J. Zheng, H. Zhou, and Z. Lu, Quantitative proteomic analysis uncovers inhibition of melanin synthesis by silk fibroin via MITF/tyrosinase axis in B16 melanoma cells, Life Sci., 284, 119930 (2021). (16) F.M. Vilela, F.M. Oliveira, F.T. Vicentini, R. Casagrande, W.A. Verri Jr., T.M. Cunha, and M.J.V. Fonseca, Commercial sunscreen formulations: UVB irradiation stability and effect on UVB irradiation- induced skin oxidative stress and inflammation, J. Photochem. Photobiol. B, 163, 413–420 (2016). (17) X. Yang, F. Peng, J. Huang, Z. Chen, and J. Zhang, Particulate matter 2.5 induced hyperpigmentation in reconstructed human epidermis model (MelaKutis®), Chin. Med. J. (Engl.), 135(4), 502–504 (2022). (18) M.J. Randall, A. Jüngel, M. Rimann, and K. Wuertz-Kozak, Advances in the biofabrication of 3D skin in vitro: healthy and pathological models, Front. Bioeng. Biotechnol., 6, 154 (2018). (19) J. Qiu, M. Chen, J. Liu, X. Huang, J. Chen, L. Zhou, J. Ma, P. Sextius, A.M. Pena, Z. Cai, and S. Jeulin, The skin-depigmenting potential of Paeonia lactiflora root extract and paeoniflorin: in vitro evaluation using reconstructed pigmented human epidermis, Int. J. Cosmet. Sci., 38(5), 444–451 (2016). (20) S. Goenka and S.R. Simon, A novel pro-melanogenic effect of standardized dry olive leaf extract on primary human melanocytes from lightly pigmented and moderately pigmented skin, Pharmaceuticals (Basel), 14(3), 252 (2021). (21) W. Karunarathne, I.M.N. Molagoda, S.R. Park, J.W. Kim, O.K. Lee, H.W. Kwon, M. Oren, Y.H. Choi, H.W. Ryu, S.R. Oh, W.S. Jo, K.T. Lee, and G.Y. Kim, Anthocyanins from Hibiscus syriacus l. inhibit melanogenesis by activating the ERK signaling pathway, Biomolecules, 9(11), 645 (2019). (22) S. Goenka and S.R. Simon, Asoprisnil, a selective progesterone receptor modulator (SPRM), inhibits melanosome export in B16F10 cells and HEMn-DP melanocytes, Molecules, 25(16), 3581 (2020). (23) H. Choi, I. Young Ryu, I. Choi, S. Ullah, H. Jin Jung, Y. Park, Y. Hwang, Y. Jeong, S. Hong, P. Chun, H. Young Chung, and H. Ryong Moon, Identification of (Z)-2-benzylidene-dihydroimidazothiazolone derivatives as tyrosinase inhibitors: anti-melanogenic effects and in silico studies, Comput. Struct. Biotechnol. J., 20, 899–912 (2022). (24) L. Zhou, P.P. Huang, L.L. Chen, and P. Wang, Panax notoginseng saponins ameliorate aβ-mediated neurotoxicity in C. elegans through antioxidant activities, Evid. Based Complement. Alternat. Med., 2019(4), 7621043 (2019). (25) J. Tang, L. Xiong, X. Shu, W. Chen, W. Li, J. Li, L. Ma, Y. Xiao, and L. Li, Antioxidant effects of bioactive compounds isolated from cordyceps and their protective effects against UVB-irradiated HaCaT cells, J. Cosmet. Dermatol., 18(6), 1899–1906 (2019). (26) Y. Wang, W. Li, S. Xu, R. Hu, Q. Zeng, Q. Liu, S. Li, H. Lee, M. Chang, and L. Guan, Protective skin aging effects of cherry blossom extract (Prunus Yedoensis) on oxidative stress and apoptosis in UVB- irradiated HaCaT cells, Cytotechnology, 71 (2), 475–487 (2019). (27) M.J. Randall, A. Jüngel, M. Rimann, and K. Wuertz-Kozak, Advances in the biofabrication of 3D skin in vitro: healthy and pathological models, Front. Bioeng. Biotechnol., 6, 154 (2018). (28) K. Yamauchi and T. Mitsunaga, Methylquercetins stimulate melanin biosynthesis in a three-dimensional skin model, J. Nat. Med., 72(2), 563–569 (2018). (29) J.H. Hwang, H. Park, D.W. Choi, K.T. Nam, and K.M. Lim, Investigation of dermal toxicity of ionic liquids in monolayer-cultured skin cells and 3D reconstructed human skin models, Toxicol. In Vitro, 46, 194–202 (2018). (30) M. Ashooriha, M. Khoshneviszadeh, M. Khoshneviszadeh, S.E. Moradi, A. Rafiei, M. Kardan, and S. Emami, 1,2,3-Triazole-based kojic acid analogs as potent tyrosinase inhibitors: design, synthesis and biological evaluation, Bioorg. Chem., 82, 414–422 (2019). (31) M. He, M. Fan, W. Liu, Y. Li, and G. Wang, Design, synthesis, molecular modeling, and biological evaluation of novel kojic acid derivatives containing bioactive heterocycle moiety as inhibitors of tyrosinase and antibrowning agents, Food Chem., 362, 130241 (2021).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)