315 Microsponge Loaded Topical Gel A microsponge is a polymeric delivery system comprised of porous microspheres. It has a large, porous surface with small sponge-like spherical particles. It provides good stability with reduced side effects and favors modified drug release. This technology has many constructive characteristics, which makes it a unique drug-delivery vehicle (4). Microsponges are based on microscopic, polymer-based microspheres and encapsulate a wide range of drug substances that can be incorporated into various dosage forms, such as gel, cream, liquid, or powder (5). Benzoyl peroxide is effective for treating acne lesions, and it does not induce antibiotic resistance. It may be combined with salicylic acid, sulfur, erythromycin, clindamycin (antibiotics), or adapalene (synthetic retinoid). Two common combination drugs include benzoyl peroxide/ clindamycin and adapalene/benzoyl peroxide, an unusual formulation considering most retinoids are deactivated by peroxide combination products such as benzoyl peroxide/clindamycin and benzoyl peroxide/salicylic acid, which appear to be slightly more effective than benzoyl peroxide alone for the treatment of acne lesions (6). The combination tretinoin/benzoyl peroxide was approved in 2021. Benzoyl peroxide for acne treatment is typically applied to the affected areas as a gel, cream, or liquid in concentrations of 2.5% increasing through 5.0% (7). In general gels are preferred over solutions and liquids because of their semi-solid consistency, which contributes to the retention of the active substance at the site of application for a prolonged time, as well as their excellent spreading properties, cooling effect due to solvent evaporation, and aesthetically pleasing qualities (8). Considering poly lactic-co-glycolic acid’s (PLGA) natural antibacterial properties, wound-healing properties, and calming effect, as well as its biodegradable and biocompatible features, PLGA-based hydrogels might be preferred over conventional gelling agents such as cellulose derivatives. Considering this information, the aim of this study is to design and develop the formulation of a topical, semi-solid dosage form, carbopol-based gel with microsponges loaded with benzoyl peroxide and PLGA. Sorbitan monooleate is a nonionic surfactant used as emulsifying agents in the preparation of emulsions, creams, and ointments for pharmaceutical and cosmetic use. It has various types of span 20, span 40, span 60, and span 80. The sustained drug release at the site of action, reduced side effects, and frequency of application are expected to result in better efficacy of acne treatment and improved patient compliance (9). MATERIALS AND METHODS The benzoyl peroxide and PLGA were supplied from Carbanio.com. The polyvinyl alcohol (PVA) and carbopol were supplied from Sigma Aldrich (Chennai, India). The sorbitan monooleate 80, propylene glycol, methyl paraben. and propyl paraben were supplied from Nice Chemicals Pvt. Ltd (Chennai, India). DETERMINATION OF SOLUBILITY PROFILE Aqueous solubility is an important physiochemical property of drug substances as it determines systemic absorption and in turn therapeutic efficacy (10). The solubility of benzoyl peroxide in water was carried out per US Pharmacopoeia monograph. The solubility of drugs in methanol, ethanol, and chloroform was determined by adding an excess of the drug in the solvent (11).
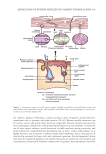
316 JOURNAL OF COSMETIC SCIENCE ULTRAVIOLET SPECTROSCOPIC METHOD FOR ESTIMATION OF BENZOYL PEROXIDE Preparation of standard stock solution. An accurately weighed quantity of 100 mg of benzoyl peroxide was taken in a 100 mL volumetric flask and dissolved by using 5 mL of methanol. Finally, the volume was made with methanol up to 100 mL to produce 1 mg/mL of solution. Scanning. A series of concentrations (i.e., 0.5, 1, 2, 3, 4, and 5 µg/mL) were prepared by using the aforementioned stock solution and scanned between 200–400 nm. The absorption maxima obtained was 235 nm and used for further studies. Preparation of calibration curve. A standard solution containing 1 mg/mL of benzoyl peroxide was prepared in methanol by dissolving 50 mg of benzoyl peroxide pure in 50 mL of methanol. From this solution, a working standard solution of concentration 0.5–5 µg/mL of benzoyl peroxide was prepared by dilution with methanol (12). The absorbance of the solution was measured at 235 nm against blank. All spectral absorbance measurements were made by using a Shimadzu ultraviolet–visible spectrophotometer (SRIHER, Chennai, India). PREPARATION AND CHARACTERIZATION OF MICROSPONGES A central composite design (CCD) consisting of three variables was used to analyze the sensitivity of the responses against the variations in the experimental variables (Figure 1). CCD comprises of 2k factorial from center, 2k points for α, and the quadratic terms used for generation of a center point. In this design, two different levels coded as +1 and −1 for factorial, central, and axial points, respectively, were investigated (13). For the three variables under investigation, the response model is depicted by the equation: N k =++=[]2 2k 14 N0 runs, where K, the number of variables, =3, N =++=[]8 6 0 14 runs. Figure 1. Preparation of microsponges (14,15). PVA: polyvinyl alcohol.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)