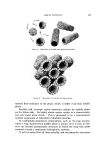
.I. •h'oc. Cosmetic Chemists, 19, 581-594 (Aug. 19, 1968) Liquid Crystals: The Mesomorphic Phases of Surfactant Compositions F. B. ROSEVEAR, Ph.D.* Presented December G, 19G7, New York City Synopsis-"Liquid crystals" are "mesomorphic" structure types between liquids, which are random, and crystals, which are periodic in all three dimensions. The mesomorphic state is surprisingly widespread in organic, and even certain inorganic, compositions. In the surfactant industry, mesomorphic material by the ton occurs as an intermediate in processing. The layered "smectic" structure of the industrially important "neat" phase will serve as an introduction to mesomorphic structures. In each layer, elongate•t molecules or other units are arranged parallel to each other and with their head ends at the interface between layers the stacking of layers is periodic. Laterally, however, the molecules have a random, liquidlike arrangement. Thus a single structure has a hybrid liquid and crystalline nature. The layers, however, slide easily over each other and the structure flows under its own weight even while maintaining crystallike periodicity. The chemical and physical factors leading to this and other mesomorphic structures in surfactant compositions are reviewed. INTRODUCTION Even those with some scientific background are likely to think in terms of just three states of matter: gas, liquid, and solid. Even if "solid" really means "crystal," one is likely to overlook the important structural types between liquid and crystal. Yet glasses, and natural and synthetic polymers, are familiar materials whose structures are intermediate between the randomness of liquids and the periodicity of crystals. "Liquid crystals" or, more descriptively, mesomorphic structures (1) (meso • between, morph • form) are among the less well-known * The Procter & Gamble Co., Miami Valley Laboratories, Cincinnati, Ohio 45239. 581
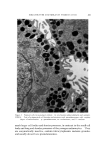
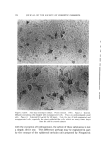
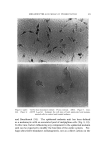
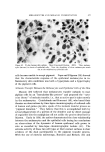
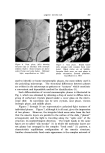
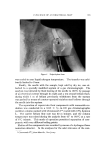
582 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS intermediate types. Yet these, too, occur in some relatively coralnon materials and in this paper the important role of these structure-types in soaps and other surfactants will be examined. NEAT PHASE Mesomorphic structures will seem less unreal and mysterious if we consider a relatively simple example: the "neat" phase of surfactant systems. This material has been known for many years as the soap- rich upper layer of the soap kettle. Still prepared by the ton., although in many cases the kettle has given way to continuous processes, neat is the starting phase for crutching, spray-drying, chilling, milling and other operations which lead to the many and varied detergent products. A notable feature of the neat phase is its relative fluidity in spite of high surfactant-content. When a tube of neat phase is inverted to a tilted position, it is seen to flow readily under its own weight. However, the flow and the surface are not smooth as with honey, but are more like those of a soft petroleum jelly. Yet the latter is a two-phase system of oil thickened with hydrocarbon crystals, whereas neat is a single phase whose flow properties arise from its structure. While neat has a yield value, it has ample fluidity to permit pumping in plant operations. The mesomorphic structure responsible for the ready flow of the neat phase is the type known as "smectic" (1). On a molecular scale, the smectic structure consists of flexible layers of equal thickness. Within each layer the molecules are (a) essentially parallel to each other, hence perpendicular to the surface of the layer, but (b) not regularly spaced in the lateral directions this arrangement has been likened by one writer to that of the people on a crowded dance floor. The lateral ar- rangement is thus liquid-like, but the stacking of the layers has a crystalline regularity manifest in sharp X-ray long-spacings. Even in this direction, however, the structure is not fully crystalline, as the in- dividual molecules in one layer do not have a definite relationship to those in the next. This allows the layers to slide readily over one an- other while maintaining their crystal-like periodic arrangement, and helps account for the relative fluidity of even concentrated surfactants in the neat phase. In a sense, the layer structure can be seen directly under the microscope in the interesting terraced droplets often exhibited by neat phase (Fig. 6). The step-heights are, of course, multiples of the smectic-layer-thickness, but they provide a striking contrast to the smooth, rounded surface of drops of water, oil and other conventional liquids.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)