STUDIES ON NONAQUEOUS EMULSIONS 633
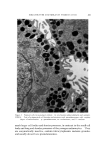
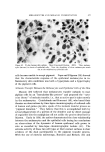
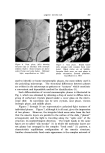
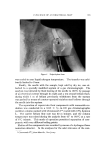
634 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Preparations which did not exhibit detectable separation after standing for this period of time were considered stable. Preparations which remained emulsified for at least one hour but showed evidence of sep- aration within seven days were classified as semistable. Those which separated immediately or within one hour after mixing were considered unstable. All procedures were conducted at a temperature of 25 • 1.0øC, except for measurement of interfacial viscosity which was conducted at 20 •= 1.0øC. RESULTS Results of attempted emulsification of propylene glycol and poly- ethylene glycol 400 with olive oil using selected surfactants are sum- marized in Table I. The results obtained with comparable systems of glycerin and olive oil are included for comparison. Interfacial tension and interfacial viscosity data are summarized in Table II. Viscosities and flow characteristics of six representative preparations are summarized in Table III. A photomicrograph of a typical oil-in-polyol emulsion is shown in Fig. 3. The emulsion contains glycerin and olive oil emulsified with 1.0% triethanolamine and stained with 1.0% each of D&C Red No. 18 and methylene blue dyes. The internal phase appears as dark-colored spheres against a light background. Air bubbles appear as heavily outlined spheres. Figure 4 is a photomicrograph of a typical polyol- in-oil emulsion. The emulsion shown contains glycerin and olive oil emulsified with 5.0% glyceryl monostearate and stained with 1.0% each of D&C Red No. 18 and methylene blue dyes. In the latter figure the internal phase appears as light-colored spheres against a dark background. CONCLUSIONS AND DISCUSSION Table I shows that combinations of glycerin and olive oil emulsified more readily than either propylene glycol or polyethylene glycol 400 and olive oil. Further, the emulsions of glycerin and oil were more stable, exhibited a wider range of physical characteristics, and required much lower levels of surfactant. It has previously been demonstrated (3) that conventional theories of emulsification, such as the HLB system of Griffin (10), are not readily adaptable to an explanation of the nonaqueous systems. For example,
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)