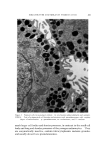
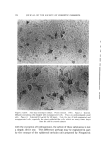
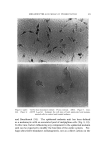
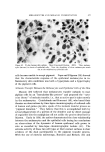
STUDIES ON NONAQUEOUS EMULSIONS 637 It has been demonstrated that stable emulsions of glycerin and olive oil can be produced with surfactant precursor levels as low as 0.6 mg ammonia or 2.0 mg 2-amino-2-methyl-l,3-propanediol (AMPD) per 100 ml of emulsion having a phase volume of 0.4 or with 0.84 mg am- monia or 2.7 mg AMPD per 100 ml of emulsion having a phase volume of 0.58 (4). These emulsions were shown through particle-size and rheological studies to be stable over a period of 60 days, and extrap- olation of the data indicates that stability will be maintained for much longer periods of time (5). It was further shown that at these low surfactant levels monolayer films ranged from 89 to 98% interfacial coverage with mean molecular area coverage in the range of 229 to 419 A •- per molecule. The unusual stability of some of the preparations probably depends on several factors such as interfacial tension, interfacial viscosity, presence of a mixed monolayer and others. The data included in Table II indicate that the addition of AMPD in the polyol phase had only a slight effect on the interfacial tension between the polyol and olive oil. It had previously been demonstrated (4) that interfacial tension measurements of glycerin-oil systems are time-dependent. In this study, drop volumes were determined as those volumes which required two minutes to separate and descend from the dropping tip. Under these conditions the changes effected by the surfactant precursor were not significant. Interfacial viscosity, on the other hand, was strongly influenced by the presence of AMPD. The existence and importance of surface viscosity (and interfacial viscosity) have been noted and studied by various investigators (11-20). The "viscous traction" method of Davies (11) and Davies and Mayers (8) was utilized by Hamill and Petersen to study the interfacial viscosity characteristics of systems of glycerin and olive oil with and without added surfactant precursor (6). Their data are included in Table II for comparison with similar studies conducted on systems containing propylene glycol and polyethylene glycol 400. An increase in interfacial viscosity and a more rigid film resulted when surfactants or surfactant precursors were present. It was noted in this study that the retardation time of the insoluble particle in the "canal" was also somewhat time-dependent. The longer the time of contact between the polyol, which contained the AMPD, and the oil, the greater was the retardation time of the insoluble particle. Systems containing propylene glycol and oil with added surfactant showed the greatest amount of time-dependency. The
638 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ten-minute interval is, therefore, arbitrary for this measurement. Thus, it may be concluded that the interfacial film serves as a mechanical barrier to coalescence and probably plays a significant role in the stability of some of the nonaqueous emulsions. In view of the fact that the quantity of free fatty acids present in the olive oil exceeded that necessary for neutralization of added amine, it is reasonable to assume that a mixed monolayer consisting of the added surfactant and intrinsic fatty acids exists at the interface. According to Osipow (21) and Schulman and Cockbain (13, 14), combinations of water-insoluble long-chain compounds and ionic surfactants produce more stable emulsions than those from ionic surfactants alone. Other factors which probably contribute to emulsion stability are the high viscosity, relative homodispersity, presence of thixotropy and small droplet size in most of the emulsions. As would be expected, the six representative emulsions presented in Table III exhibited nonNewtonian flow characteristics. The data contained in Table III indicate that all of them were pseudoplastic and thixotropic. Viscosities varied widely, as would be predicted from the wide range of physical characteristics which they exhibited. SUMMARY A departure from traditional methods and concepts of emulsion technology has resulted in the development of a family of nonaqueous emulsions having many interesting and unique properties. Liquids representing the polar phase include glycerin, propylene glycol and polyethylene glycol 400. Olive oil was used as the nonpolar phase. Emulsification was effected by means of anionic, cationic, and nonionic surfactants. The order of mixing of these components is critical. The resulting emulsions vary in physical characteristics from clear, viscous, transparent fluids to opaque, semisolid creams. In all instances, glycerin appears to be the agent of choice in producing stable, superior emulsions at low surfactant concentration. In constrast to conventional oil-water emulsion systems, the oil- polyol systems do not emulsify in accordance with predictions based on the HLB value of any given surfactant. Rather, the chemical nature of the surfactant appears to govern the type of emulsion which forms and the method of mixing. In all cases where polyol-in-oil emulsions were formed and/or the order of mixing favored addition of surfactant to the oil phase (Method II or IIa) the surfactant was a derivative of stearic acid.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)