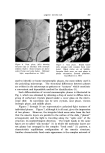
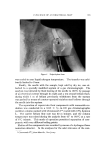
LIQUID CRYSTAI. S b91 THE SYSTEM:SODIUM PALMITATE-WATER TEMP.•C, NEAT 11•1-•-- / "• N ] SU•NEATr• ',• / •]• •,. A (ISOTROPIC) /• SUPE RWA XY•-• • -'t NEAT F•uu • • + • (NEAT I) • •- PHASE I' •u•w•xY-• • •'• I + -- • '• I00 80 60 40 •0 % SODIUM PALMITATE • Figure 16. A composite phase diagram of sodiron palmitate-water compiled from several sources (10-8) 80[ THE SYSTEM: TALLOW SOAP-NoCI-H20 , IO0*C A = NEAT SOAP 60 B = NEAT-MIDDLE C = NEAT-MIDDLE-NIGRE % D = MIDDLE SOAP SOAP E = MIDDLE-NIGRE F- NIGRES OR SOAP SOLUTIONS 40 G = NEAT- NIGRE H = NEAT-NIGRE-LYE K = NEAT-LYE N = NIGRE- LYE 20 2 4 6 8 I0 12 % NoCI Figure 17. •hree-component soap-water- salt diagram history of "liquid crystals" with such compounds as p-azoxyanisole and ethyl p-azoxybenzoate. Figure 17 shows the effects of adding NaC1 as a third component (14). On addition of NaC1 at relatively low surfaetant concentration (a few per cent) there is at first no change from homogeneous isotropic liquid. At several per cent N aC1, the solution disproportionates into two isotropic layers, one somewhat richer in soap. As NaC1 content increases, the two layers gradually separate farther in composition. Since the two layers are really different compositions in the same iso- tropic liquid phase, some means of differentiating them is needed. The phase-name for the less soapy layer is "lye" because in the early stages of soap-boiling, the electrolyte is actually a soda-lye. The phase-name for the soap-richer layer is "nigre," referring to the accumulation of the coloring matter and glycerine in this layer in soap-boiling. When sufficient salt is added to these layered compositions, a long, narrow, three-phase triangle (neat/nigre/'lye) is entered in this triangle the composition of the three phases is fixed and, on settling, the topmost phase is neat. On the high-NaC1 side of this triangle, neat and lye coexist until, when sufficient NaC1 is present, the lye reaches saturation and additional NaC1 remains crystalline in the neat/lye/solid-NaC1 triangle. If NaC1 is now introduced at higher surfaetant levels, e.g., about
592 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 40% where middle phase is present in the absence of NaC1, middle phase dissolves progressively until with several per cent NaC1 only a con- centrated isotropic solution remains middle phase has been "salted in." Additional NaC1 now "salts out" neat phase and the two-phase neat/nigre region is entered. Here the compositions of upper and lower layers vary with total composition but, for a given total composition, are fixed at the ends of the appropriate tie-line. With further NaC1 the long, narrow neat/nigre/lye triangle, encountered earlier, is entered but now the proportion of the neat layer is higher. Soap-boiling oper- ations are carried out close to this three-phase triangle but nearer the neat-phase end. Kettle-finishing is done in the triangle or on the neat/ nigre side and the settled neat phase is relatively free of entrained iso- tropic solution. On the other side of the triangle, in the neat/lye region, the consistency of the neat is grainy or curdly with much entrained lye- phase. This is desirable for replacing residual caustic with salt but requires hydrating back into the three-phase triangle or the neat/' nigre region for finishing as a smooth, homogeneous neat layer. Free fatty acid and fatty alcohol have an effect somewhat similar to that of sodium chloride on the soap phases. For example, addition to a soap-water middle phase suppresses middle and in sufficient quantity leads to a layer of neat phase. Formulators will be interested in some generalizations on the effect of composition variables on the occurrence of mesomorphic phases. All three types of surfactants---anionic, cationic, and nonionic--exhibit mesomorphic phases. Soaps, alkyl ammonium halides (15), alkylamine oxides (16), and polyoxyethylene esters and ethers are representative. Among the soaps, the mesomorphic phases of the unsaturated soaps, such as the oleate, occur closer to room temperature than do their sat- urated counterparts this is true also of the shorter-chain soaps, such as the laurate. In general, the sodium soaps require heating for attain- ment of mesomorphic phases. In the potassium, ammonium and tri- ethanolammonium soaps, however, the mesomorphic phases occur at or near room temperature.* PRACTICAL APPLICATIONS OF MESOMORPHIC COMPOSITIONS The practical applications of the neat phase have been in processing rather than in formulations for sale, although it has been marketed in a * Because of the existence of mesomorphic phases of potassium soaps at ordinary tempera- tures, the making of the early soaps by the action of wooct-ash leachings, largely potassium carbonate, led to mesomorphic soft soap.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)