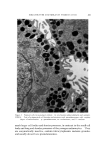
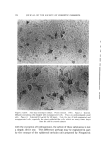
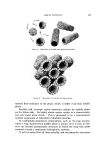
LIQUID CRYSTALS 583 Figure 1. Structure of neat phase Figure 2. Structure of middle phase When surfactants exist in a smectic structure, as in the neat phase, molecules in adjacent layers alternate in direction. If water is present, the polar heads of adjacent layers share the same aqueous interface (Fig. 1') this means that the nonpolar tails of molecules in adjacent layers similarly share a common nonpolar environment (2-4). It will be seen that all of the mesomorphic surfactant structures show such polar and nonpolar zones. MIDDLE PHASE A second important mesomorphic phase in surfactant/water systems is the "middle" phase. In spite of a substantially higher water-content, middle phase is much stiffer than neat. Middle does not flow under the influence of gravity, though it does flow plastically if subjected to a sufficient external force. This stiffness renders pumping impossible and, coupled with an inherent translucency, gives rise to the term "gum soap." It is easy to picture the consternation caused by even partial conversion of a kettle-full of neat phase to middle because of inadvertent overhydration. The stiffness of middle phase lies in the absence of a smectic layer- structure. Its structure is shown in Fig. 2. The basic unit of this structure can be thought of as neat-phase layers rolled up into cylinders. Normally, the hydrophili½ heads of the molecules are on the outsides of these cylinders and the tails comprise a hydrocarbon interior. These * Figures 1-5, courtesy of K. D. Lawson, Miami Valley Laboratories.
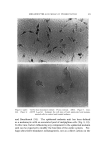
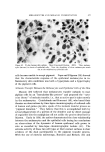
,584 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS cylindrical aggregates, approximately two molecules in diameter and o[ indefinite length, are aligned parallel to each other in bundles having a hexagonally packed cross-section (2-4). The bundles are thus crystalline in two dimensions however, since the component cylinders are not in register longitudinally and are possibly of assorted lengths, the bundles are liquid-like in their long direction. Thus, whereas neat phase is liquid in two dimensions and crystalline in one, middle phase is liquid in one dimension and crystalline in two. The latter type of structure was first demonstrated (5) in a meso- morphic phase of tobacco mosaic virus whose molecules, like the mo- lecular aggregates of middle phase, are elongated cylinders. It should be noted that this structure was among a number of theoretically possible mesomorphic structures deduced by C. Hermann (6, 7) in 1•,t31 on the basis of the three types known at that time and named smectic, nematic and cholesteric by Friedel (1). The neat and middle structures, each of which has one unique structural direction, are in the class of "anisotropic" structures whose properties vary with direction. Possibly the most strikin• evidence of this anisotropic character is the double refraction, or birefringence, by which the sample appears bright when viewed between two light- polarizers arranged in the so-called "crossed" position. Isotropic materials, such as ordinary solu•rions and those crystals which possess no unique direction, appear dark between crossed polarizers. OTHER NON-CRYSTALLINE PHASES If any of the mesomorphic phases are sufficiently diluted, they dis- solve to form an isotropic solution. Usually middle phase is the im- mediate precursor of this solution in this case one may thin. k of the soap cylinders moving sufficiently far apart that the hexagonal bundles disintegrate, leaving the individual cylinders randomly distributed as "cylindrical micelies" (Fig. 3). On further dilution, the cylinders be- come shorter, until at some fairly high dilution they degenerate into spheres, a geometrical shape of minimum surface energy for a given volume. Below a certain concentration, the spherical micelles disappear entirely to leave only molecularly dispersed surfactants. In addition to neat and middle, and usually at compositions between those phases, some surfactants exhibit a third mesomorphic phase which is actually isotropic. Its structure is interpreted as a face-centered cubic arrangement of spherical micelles with water filling in the voids (3, 4) (Fig. 4). The close packing of the micelles accounts for the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)