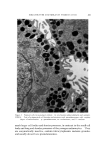
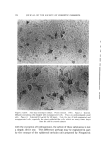
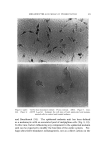
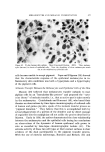
STUDIES ON NONAQUEOUS EMULSIONS 639 Studies on emulsion stability, based on viscosity changes and droplet size growth over a 60-day period, revealed that stable emulsions result even when exceedingly low concentrations of surfactant precursor are employed (as low as 0.6 mg ammonia per ll)0 ml emulsion). Based on the desirable physical characteristics and stability prop- erties of these emulsions and the inherent values of the components which they contain, it appears that these preparations can be used c•dvantagcously in cosmetic technology. (Received December 8, 1967) (14) (15) (16) REFERENCES (1) Pharn•acopeia of lhe United States, 17th Rev., Mack Publishing Co., Easton, Pa., 1965, p. 786. (2) McMahon, J. D., Hamill, R. D., and Petersen, R. V., Emulsifying effects of some ionic surfactants on a nonaqueous immiscible system, J. Pharm. Sci., 52• 1168 (1963). (3) Petersen, R. V., Hamill, R. D., and McMahou, J. D., Emulsifying effects of some non- ionic surfactants on a nonaqueous immiscible system, Ibid., 53, 651 (1964). (4) Hamill, R. D., Olson, F. A., and Petersen, R. V., Some interfacial properties of a non- aqueous emulsion, Ibid., 54, 537 (1965). (5) Hamill, R. D., and Petersen, R. V., Effects of aging and surfactant concentration on the rheology and droplet size distribution of a nonaqueous emulsion, Ibid., 55• 1268 (1966). (6) Hamill, R. D., and Petersen, R. V., Effect of surfactant concentration on the inter- facial viscosity of a nonaqueous system, Ib•d., 55, 1274 (1966). (7) Adamson, A. W., Physical Chemistry of Su(l•zces, Interscience Publishers, Inc., New York, X. Y., 1960, Table 1-4, p. 25. (8) Davies, J. T., and Mayers, G. R. A., Studies on the interfacial viscosities of mono- layers, Trans. Faraday Sot., 56, 691 (1960). (9) Runikis, J. O., Hall, N. A., and Rising, L. W., A note on the use of the Brookfield RVT viseometer with thickening agent dispersions, J. Am. Pharm. Assoc., Sci. Ed., 47, 758 (1958). (10) Griffin, \V. C., Classification of surface active agents by HLB, J. Soc. Cosmetic Chemists, 1,311 (1949). (11) Davies, J. T., Seco•zd Dzler•mtional Congress of Surface Activity, Vol. I, Schuhnan, J. H., ed., Buttersworth Scientific Publications, London, England, 1957, p. 220. (12) King, A., Some factors governing the stability of oil-in-water emulsions, Trans. Fara&•v Sot., 37, 168 (1941). (13) Schuhnan, J. H., and Cockbain, E.G., Molecular interactions at oil/water interfaces [. Molecular complex formation and the stability of oil in water emulsions, Ibid., 36• 651 (1940). Schuhnan, J. H., and Cockbain, E. G., Molecular interactions at oil/water interfaces II. Phase inversions and stability of water-in-oil emulsions, Ibid., 36,661 (1940). Becher, P., Theoretical aspects of emulsification, a background for cosmetic formula- tion, Am. Perruiner Cosmetics, 77, 21 (1962). Shotten. E., and White, R. F., Stabilization of emulsions with gum acacia, in Rheology oj' Emuisio•ts, Shernmn, P., ed., Pergamon Press, New York, 1963.
640 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (17) Sunruer, C. G., The Theory of Emulsions and Their Technical Treatmcnl, The Blakiston Co., New York, 1954. (18) Blakey, B.C., and Lawrence, A. S.C., The surface and interfacial viscosity of soap solutions, Disc. Faraday Sot., 18, 268 (1954). (19) Taubnmn, A. B., and Koretkii, A. F., The importance of the structure-mechanical fac- tor in the stability of emulsions, Kolloid Zh., 20, 676 (1958) through Chem. Abstr., $3, 11948e (1959). (20) Rehbinder, P., and Taubman, A., Structures colloidales dans les solutions et les couches d'adsorption des detergents leur relation to la stabilisation des systemes dis- perses, Proc. Third Intern. Congr. Surface Activity, 1, 209 (1960). (21) Osipow, L., Surface Chemistry, Reinhold Publishing Corp., New York, 1962, p. 333.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)