INFLUENCE OF ANTIBACTERIAL SOAP (113 primary odor and secondary odor? What are the variations in these effects among individuals ? To what extent are the effects interrelated ? How long after discontinuing the use of the medicated soap do the e[- fects last ? EXPERIMENTAL PROCEDURES One method for evaluating the odor-reducing effect of antibacterial soaps by the in vivo panel technique involves 40 to 60 panelists. Each must be capable of distinguishing differences in odor of the underarm portions of his or another's T-shirt. The results obtained by a panel are not necessarily pertinent to the odor actually in the axillae this fol- lows from the distinction of conditions prevailing in the primary sites cf axillary odor (in axillae) and those in the secondary sites of odor (in the garment). The primary odor originates in the axillae itself from the action of gram-positive bacteria on apocrine sweat (4). The precursors of this odor are the original organic compounds contained in the apocrine sweat. The secondary odor develops on the garments in contact with or in the vicinity of the axillae. The bacterial growth environment in the secondary odor sites differs from that in the axilla in humidity, tem- perature, degree of aeration, relative composition of nutrients, and, in the case of skin care with antibacterial agents, in the relative concentra- tion of the agent. Therefore, primary axillary odors can differ from secondary axillary odors in intensity and quality. Strong secondary odor does not necessarily indicate that the primary odor is strong also. Hence, for a full evaluation of the effects, both secondary and primary odors must be evaluated. Selection of Subjects and Schedules Four young healthy Caucasian males participated as volunteer sub- jects in the study. Water generation rates, bacterial population density on axillary skin, and odor intensities from secondary sites were measured by methods outlined in the subsequent section. In addition, for three of the subjects odor intensities were rated by the gas chromatographic rating method also described in the subsequent section. The subjects washed the axillae with the assigned soaps every morn- ing except on days when test samples were collected. On these days, the intensity of the odor from the secondary source was rated by a panel of three experienced judges. Then axillary vapor samples were taken and, within 30 to 60 minutes, the sample for the bacterial count was col-
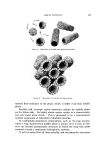
614 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS lected. A supervised wash with the scheduled soap then ensued. There were two sample collection mornings per week. The subjects were kept on the neutral soap for several weeks. Then the medicated soap was used for two or more weeks. Finally, the medicated soap was discontinued, and samples were taken as usual the next morning, before washing with the neutral soap. On the subsequent two mornings, no soaps were used. Thus, the last two samples in the series were taken 48 hours and 72 hours after the last use of the medi- cated soap, with an intervening neutral soap wash at 24 hours. These last two samples in each series were taken so as to determine whether odor control lasts for a few days after discontinuing the use of the medi- cated soap. Vapor Collection and Separation Techniques By the use of dental impression techniques combined with heat- forming of polyethylene sheet, polyethylene forms were prepared that fitted the axillary surfaces of the subjects (Fig. 1). The forms, when attached to the axillae by shoulder straps, formed a cavity 10 to 20 cc in volume at the apex of the axillary vault and sealed the edges of the cavity by gently pressing against the surrounding skin areas. There- fore, the axillary surface was reproducibly defined for each axilla of the same subject. Tubing connected to the polyethylene form permitted (a) introduc- tion of high-purity cryogenically purified helium (60 cc/minute) and (b) removal of vapor-laden helium. The helium was removed through a heated Teflon tube into a spiral glass tube thermostated at 30øC and coated internally with high-purity Apiezon L* grease film. Organic vapors dissolved in the grease, while water vapor with helium passed through. Water vapor was collected in a small dry ice-cooled condenser for weighing. Flow rates of helium entering the axilla and leaving the spiral glass tube were monitored to correct the seating of the form on the axilla if a helium leak was observed. The duration of collection was usually 30 minutes. Between tests, the polyethylene forms were washed with acetone and stored at 50øC in a continuously pumped vacuum desiccator to keep the forms free of organic vapors. The organic components of the vapors, trapped in the glass spiral, were transferred by a high-purity helium to a special stainless steel needle. During transfer, the spiral was heated to 80øC, while the needle * Apiezon L ©, Apiezon Products Limited, England.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)