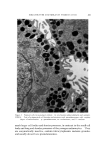
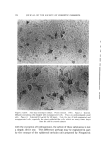
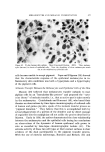
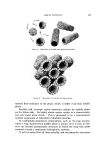
STUDIES ON NONAQUEOUS EMULSIONS 629 factant precursor was added to the olive oil. The polyol was then added in small portions, with rapid trituration after each addition. As an alternative to trituration, a Waring blendor equipped with a Polytron Rotor-Stator* was employed in the emulsification procedures. In Method Ia the surfactant was added to the polyol contained in the blendor. The requisite amount of oil was then added as a slow con- tinuous stream during attempted emulsification. In Method IIa the blendor was again utilized but the order of mixing was reversed as in Method II. All reagents, except glycerin, were USP quality and were used without further purification. The glycerin was 99+% purity, confirmed by its refractive index. Appropriate concentrations of ammonia were pre- pared from stock solutions, by weight, of the anhydrous gas contained in the polyol or oil. Deaeration of some of the emulsions was carried out while stirring in vacuo. Because the emulsions could not be visualized by ordinary microscopic methods, a Wild phase contrast microscope, illuminated by light at 475 m• and providing 480X magnification, was utilized in studying and in preparing photomicrographs of the emulsions. Emulsions were also prepared which contained 1.0% D&C Red No. 18 dye in the oil phase and/or 1.0% methylene blue in the polyol phase. Emulsions prepared in this manner could be visualized by ordinary microscopic methods. However, the dyes had a pronounced effect on emulsion stability and could not be used in preparations containing anionic surfactants or in preparations containing low concentrations of sur- factants. Emulsion type was confirmed by the phase-dilution method. Interfacial tension measurements were performed with a drop- volume tensiometer (Fig. 1) specially constructed for this and previous studies. Interfacial tension is calculated as a function of drop volume by means of equation (1): •(p - po)• V = f2•rr (1) In this equation, -• is interfacial tension, u is the volume of the drop, p -- p0 is the density difference of the two liquids, g is gravity, and f is a correction factor (7), varying with the drop volume and radius, r, of the metal tip (r/v•). * Model BEW-15 attachment for Waring blendor.
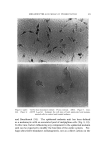
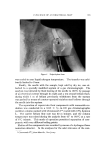
63O JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Figure 1. Drop-volume tensiometer Interfacial viscosities were measured by means of the instrument shown in Fig. 2. This instrument is a modification of the "viscous traction" viseometer of Davies and Mayers (8) and was also specially constructed for these studies. Retardation of an insoluble particle in the "canal" formed by the two concentric rings positioned at the interface of the two liquids as they revolve on the turntable is a function of the interfacial viscosity and can be expressed as surface poise by application of the graphic methods devised by Davies and Mayers. Systems were allowed to stand for ten minutes prior to measuring rotational time. Further details concerning the specifications, calculations and use of the interfadal tensiometer and interfacial viscometer are given in previous publications (4, 6). A Brookfield LVT viseometer* was employed in all viscosity mea- * Brookfield, Synchro-Lectric, Model LVT.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)