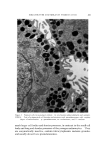
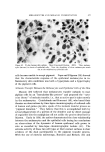
INFLUENCE OF ANTIBACTERIAL SOAP 615 .. . . :•-•-: 2- .. . Figure 1. Polyethylene form was cooled to near liquid nitrogen temperature. The transfer was arbi- trarily limited to 1 hour. Finally, the needle with the sample, kept cold by dry ice, was at- tached to a specially modified septurn of a gas chromatograph. The analysis was initiated by flash heating of the needle to 180øC by passage of an electrical current through its walls and a ten-second helium flush, during which 1 cc of helium previously withdrawn from the septurn was pushed bv means of a motor-operated stainless steel bellows through the needle into the septurn. The separation of vapors into their components with reasonable res- olution was conducted in a 10-ft X •/• -in-OD gas chromatographic partition column packed with Chromosorb P* coated with 10% Apiezon L. The carrier helium flow rate was 60 cc/minute, and the column temperature was raised during the analysis from 45 ø to 180øC at a rate of 6øC minute. This mode of operation permitted separation of com- ponents with very different boiling points. Elution of the components was recorded by means of a hydrogen-flame ionization detector. In the analyses for the odor relevance of the corn- * Chromosorb P©, Johns-Manville, New Jersey.
616 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ponents, the effluent gas from the gas chromatograph was split in half. One-half was delivered to the detector, and the other half was delivered to a sniffing port, where trained perfumers observed and classified the components as "odorous" or "nonodorous" as well as noted the nature of the odors of the odorous peaks. Odor-Intensity Measurements Two methods of odor-intensity measurements were used. In the conventional method, the axillary portion of a sweat shirt worn by the subject for a controlled amount of time was judged for secondary odor intensity by three experienced judges on a 3-point scale (2 = slight, 4 = moderate, 6 = strong) the rating was an average of their judgments. The odor-intensity rating of the primary axillary odor was obtained with the assistance of gas chromatograms. In this method, odor rele- vance of the components represented by peaks was established by judging which peaks are odorous. The vapor collection, transfer and sampling procedures were dimensioned so that if a peak is found to have no odor, it was present in the initial vapor mixture at a concentration considerably below its own odor threshold and hence probably did not contribute significantly to the intensity or the nature of the odor of the initial vapor mixture. All peaks that had odors were considered poten- tial contributors to the full odor, regardless of their odor qualities. The contribution of each peak to the total odor intensity was then con- sidered proportional to 0.55 power of its gas chromatographic peak area (which is proportional to the concentration of the components in the initial sample). These relations are derived from Stevens' law of odor intensities' Intensity = k• [n•- (ni)thr]x* Here k• and X• are coefficients specific to each odorant, (n•)t•r is the odorant's odor threshold concentration, and nl is the odorant's con- centration in the gas sample delivered to the nose. The underlying simplifying assumption is that some average k and X values can be applied to all odorous peaks from the particular source. The total odor intensity is then computed as the sum of scaled intensities of all odorous peaks. This intensity, termed "gas chromatographic odor intensity," was found to correlate over a broad range reasonably well with panel- rated intensities from the same source. It was consequently used as a measure of the odor intensity of the vapors sampled and analyzed. In this text it applies to the odor from the axilla as sampled by the helium flush method.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)